Myocardial Bridges (Tunneled Epicardial
Coronary Artery)
The coronary arteries may dip into the
myocardium for varying lengths and then reappear on the heart's
surface (Figs. 212-1 to 212-9). The muscle
overlying the intramyocardial segment of the epicardial coronary
artery is termed a myocardial bridge (MB) and the artery coursing
within the myocardium is called a tunneled artery (see Figs.
212-1 to 212-3). Tunneled coronary arteries have long been
recognized anatomically, but suggested associations between
myocardial ischemia and myocardial bridges have heightened their
clinical relevance.
Tunneled coronary arteries have been presumed congenital in
origin. At least three factors have been postulated to account
for differences between the high frequency of tunneled major
coronary arteries observed at necropsy (5-86 percent and the
lower frequency of tunneled coronary arteries observed angiographically
(0.5-12 percent) Fig. 212-11 or associated
with symptoms of myocardial ischemia (18 percent):
(1) Length, thickness,and location of the
tunneled coronary segment (MB), the presence of left ventriclar
hypertrophy, hypertrophic cardiomyopathy, and aortic stenosis.
The length of the coronary arterial narrowing markedly influences
coronary hemodynamics, explaining why relatively long bridges
are present in symptomatc patients. Ultrasound studies show
that the thicker bridges are most frequent in symptomatic patients.
(2) Degree of systolic compression.The location and orientation
of the muscle bundles over the imbedded coronary artery also
affect systolic compression. Some are superficial and others
are deeper surrounding the mid or even the proximal LAD. Progressive
increases in left ventricular wall tension with the preceding
mentioned diseases may play a part in producing symptoms.
Increased intracoronary Doppler flow velocities.
As shown in Table 1 below, resting average
peak-flow velocity (APV) and average diastolic peak flow velocity
(ADPV) were higher within the bridged segment than in the corresponding
proximal and distal segments. The flow velocity increase within
the MB was bridged segment as compared to the proximal and distal
segments. Mean diastolic/systolic flow velocity ratio (DSVR)
at rest ranged from 2.4 to 2.9. Thus, intracoronary Doppler
flow measurements reveal a significant increase in APV, ADPV,
and MPV within the MB at rest, with only minor changes in systolic
flow. These flow velocity abnormalities are very consistent
across the different studies.
During rapid atrial pacing (Table 2 below),
all flow velocitieswithin the MB were increased. The flow acceleration
was largest for MPV and again smallest for ASPV within the bridge
segment. There was a significant increment in DSVR within the
MB during tachycardia, but no change proximally or distally.
Thus, atrial pacing induced a further acceleration in flow velocities
within the bridged segment, whereas the flow velocities proximal
and distal to the bridge remained unchanged. A significant increase
in DSVR appears to be the most prominent flow alteration within
the MB during stress.
Finally, qualitative analysis of the Doppler
flow profile shows a highly characteristic pattern in approximately
90% of the patients with an abrupt early diastolic flow acceleration,
which has been termed the "finger tip" phenomenon,
a rapid mid-diastolic deceleration and mid-to-late diastolic
plateau (Fig. 212-12). A retrograde
flow phenomenon during systole was detected in the majority
of patients at the proximal entry site of the bridged segments.
Atrial pacing led to further accentuation of the velocity profile,
with shortening of the diastolic plateau as well as accentuation
of the retrograde flow phenomenon.
Schwarz et al. (13) quantitatively analyzed consecutive stop-frame
images from a complete heart cycle in 14 patients and correlated
their results with Doppler flow measurements obtained in the
same patients at an identical site. There was rapid lumen reduction
during systole with the smallest diameter during late systole,
the characteristic milking effect. Early diastolic diameter
gain was clearly delayed with a persistent mid-diastolic diameter
reduction always >30%. The abrupt early diastolic flow acceleration
was due to this persistent diastolic diameter reduction. This
was followed by rapid diastolic lumen gain, which reduced the
flow velocity, and finally stabilization of the lumen diameter
leading to a plateau during late diastole. As shown by this
study, the speed of early diastolic diameter gain and systolic
compression strongly depends on heart rate, contractile state,
and peripheral resistance and may vary substantially.
CFR
The CFR, defined as the ratio of mean flow
velocity achieved at peak hyperemia to mean resting flow velocity,
was obtained after intracoronary injection of papaverine. A
normal CFR ratio should be above 3.0. Thus, CFR distal to the
bridge was abnormally reduced in all these studies, ranging
from 2.0 and 2.6 (Table 2). Impaired CFR
can be explained, besides systolic vessel compression, by the
delayed early and mid-diastolic relaxation with increased diastolic
blood flow velocities. The increase in CFR obtained after angioplasty
of the bridge segment is described in the following text.
Additional mechanisms of myocardial ischemia
Several additional factors have been shown
to influence both the degree of systolic compression and the
speed of early diastolic gain and subsequent diastolic relaxation
in patients with symptomatic MB (see effects of Length and degree
of systolic compression above and heart rate below).
(3) Heart rate. With slow rates,most coronary
blood flow is in diastole.But at higher rates the diastolic
period is shortened. In table 3 it is shown that most of the
flow disturbances occur in dastole in symptomatic patients.
Changes in systemic arterial pressure and coronary perfusion
can also singnificantly affect the severity of systolic and
consequently of early and mid-to-late diasltolic compression
in patients with MB. For example, sublingual or intracoronary
nitroglycerine and sodium nitroprusside increase the coronary
narrowing in patients with MB, whereas noradrenaline, phenylephrine,and
ergonovine decrease it.
Longer tunneled segments of coronary arteries,
more severe systolic diameter narrowing of the tunneled segment,
and tachycardia may contribute to the production of myocardial
ischemia with myocardial bridging (see Figs.
212-7 and 212-8 and myocardiobridge-table
1 and table 2). The length of coronary
tunneling may not always be an important factor in causing myocardial
ischemia, since three patients with left main intramyocardial
tunneling of greater than 40 mm have been described without
evidence of myocardial ischemia (Fig. 212-9).
Clinical symptoms and objective signs of
myocardial ischemia.
Myocardial bridges can be an incidental finding
at the time of coronary angiography. Conversly, a wide variety
of clinical syndromes including unstable angina, acute myocardial
infarction (AMI), life-threatening cardiac arrhythmias, and
sudden cardiac death have been associated with MB .
Typically, these patients are predominantly
males, 5 to 10 years younger than patients with symptomatic
coronary disease, and they have fairly severe anginal symptoms.
Typical angina is present in 55% to 70% of the cases, and atypical
angina is often reported in association with rest angina.
The mean time interval between onset of symptoms
and coronary angiography is >18 months, and there were an
average of 2.5 previous hospital admissions for angina or suspected
AMI. Some patients had documented prior anterior or septal non-Q
wave AMI. Except for 12 patients (18%) of Ge et al. who had
significant stenoses in the proximal segment of the bridged
artery, all these selected patients had isolated MB with >50%
systolic lumen diameter reduction, without angiographic evidence
of significant coronary atherosclerosis or left ventricular
hypertrophy.
Symptom-limited exercise electrocardiograms were performed in
73% to 100% of patients. As a rule, patients not undergoing
stress testing before cardiac catheterization had unstable symptoms.
Significant ischemic ST-segment depression >0.1 mV in the
anterior leads was identified in 28% to 67% of patients in whom
the test was done. Myocardial scintigraphy was obtained in 38%
to 100% of patients. Stress-induced perfusion defects of the
anterior wall or septum were documented in 33% to 63% of the
patients in whom the myocardial scan was performed.
Therefore, compared to patients with significant
singlevessel coronary disease, these selected patients with
isolated symptomatic myocardial bridging and >50% systolic
lumen diameter narrowing of the LAD probably exhibit a comparable
severity of anginal symptoms and of noninvasive signs of myocardial
ischemia. This probably also includes a similar incidence of
prior or recent unstable angina and AMI (acute myocardial infarction).
Finally, the long-term outcome of these patients may also be
as favorable as that of patients with single-vessel coronary
disease or even better, because, in contrast to atherosclerosis,
little late progression is to be expected.
Invasive diagnostic methods.
Quantitative coronary angiography, usually
in the plane showing the most severe narrowing, objectively
measures the lumen diameter reduction within the bridge segment
as compared to the proximal and distal segments and determines
the percent MLD (mean lumen diameter) narrowing both during
systole and during mid-to-late diastole. As a rule, a significant
milking effect shows an approximate 70% MLD reduction during
systole and an approximate 35% MLD reduction during mid-to-late
diastole ( Myocardiobridge -Table 1a).
The severity of the systolic and mid-to-late diastolic MLD narrowing
can be confirmed by IVUS, which has the advantage of assessing
lumen area instead of only diameter in an invariably nonspherical
vessel lumen. Ge et al.have described a highly specific "half
moon" phenomenon surrounding the MB in all 62 patients
in whom the IVUS catheter could be passed through the bridge
segment (Figure 212-10). This ultrasound
phenomenon is only found in the bridge segment and not in the
proximal or distal segment or in other coronary arteries. In
some cases where the specific "half moon" phenomenon
was demonstrated but the typical angiographic milking effect
was not obvious presumably because of only slight myocardial
bridging, the milking effect could be provoked by intracoronary
administration of nitroglycerin.
Intracoronary Doppler flow velocity measurements reveal another
highly, characteristic diagnostic pattern in patients with MB.
In approximately 90% of cases, the flow velocity curve within
the bridge segment is characterized by an early diastolic "finger
tip" phenomenon, followed by a rapid mid-diastolic deceleration
and a mid-to-late diastolic plateau (Figure
212-11). The typical "finger tip" phenomenon is
produced by an abrupt early diastolic flow acceleration as a
result of delayed vessel relaxation within the bridge segment.
As a result of the systolic compression, there is also a markedly
reduced to absent antegrade systolic flow and quite often a
typical local systolic retrograde flow phenomenon directly proximal
to the more severe site of the MB. Rapid atrial pacing accentuates
these characteristic early diastolic and retrograde systolic
flow phenomena, while it also shortens the mid-to-late diastolic
plateau (Figure 212-11). Typically,
intracoronary Doppler flow velocities at rest, especially APV
(average peak flow velocity), ADPV, and MPV (maximal peak flow
velocity) should be higher in the bridge segment than in the
proximal and distal segments and they should be accentuated
by rapid atrial pacing (Myocardiobridge -Table
1b and Table 2). Finally, a <3.0
CFR(coronary flow reserve) distal to the MB obtained after intracoronary
administration of papaverine reflects a hemodynamically significant
mid-to-late diastolic coronary, flow reduction as a result of
the increased resistance produced by the MB (Myocardiobridge
-Table 1b).
Medical treatment
Medical treatment of symptomatic, clinically
recognized myocardial bridges includes optimal doses of beta
and calcium-channel blockers (control of tachycardia and antispasmodic
effects) and antiplatelet agents with the objective of relieving
symptoms and signs of myocardial ischemia and /or protecting
against the risk of future coronary events. Negative inotropic
blockers and chronotropic agents, especially beta-adrenergic
receptor blockers, can reduce external vessel compression by
lowering systemic and intramural pressures and can improve coronary
prefusion by prolonging the period of diastole. Calcium channel
blockers are especially useful if there is a contraindication
to beta-blocker therapy or as primary therapy when coronary
vasospasm is suspected. Nitrates have been used effectively
in some patients, possibly because of their capability to reduce
preload and to relieve vasospasm. Limitation of strenous physical
activity is usually advised to avoid the deletrious effect of
tachycardia.
Percutaeous Coronary Intervention (PCI)
Stables et al. first demonstrated, in 1995,
that intracoronary stent implantation could achieve internal
stabilization of the coronary artery lumen against external
compression in a patient with a muscular bridge. In 1997, Klues
et al. reported hemodynamic, angiographic, and IVUS data immediately
and seven weeks after successful coronary stent implantation
in three symptomatic patients. Stent placement abolished the
phasic lumen compression, the diastolic flow abnormalities,
and clinical symptoms. The CFR improved from 2.4 ± 0.5
to 3.8 ± 0.3. Coronary angiography after seven weeks
revealed an identical lumen enlargement without any systolic
or diastolic diameter reduction and a further increase in CFR.
Intravascular ultrasound revealed no signs of neointimal proliferation
within or proximal or distal to the stented segments. All patients
reported remarkable clinical improvement, with increased physical
activity. Favorable short-term clinical and angiographic results
after coronary stenting were also reported by others.
Haager et al. recently described successful
coronary stenting in 11 patients who subsequently underwent
repeat coronary angiography at seven weeks and six months as
well as clinical follow-up at two years. Immediately after stent
deployment, quantitative coronary coronary angiography (QCA)
showed absence of systolic compression, and all flow abnormalities
including abrupt early diastolic flow acceleration, rapid mid-diastolic
deceleration and mid-to-late plateau, and retrograde flow during
systole were normalized. At seven weeks, QCA showed in-stent
restenosis in 5 of 11 patients (46%). Four underwent repeat
target lesion revascularization; two received coronary angioplasty
and two underwent coronary bypass surgery with an internal mammary
artery graft to the LAD. At two years, all remained free of
angina and cardiac events. As stated by the investigators, the
incidence of in-stent restenosis in these patients was not different
from that of lesions of 25-mm length and relatively small-vessel
calibers in coronary patients (Table 1a).
Long-term studies are necessary to evaluate the stability of
stent geometry, in-stent restenosis, and clinical outcome in
these patients.
Surgical treatment.
Before the current era of coronary stenting,
surgical myotomy was regarded as perhaps the treatment of choice
for patients with persistent symptoms despite intensive medical
therapy. Indeed, cleavage of the overlying muscle fibers eliminates
the phasic compression of the coronary vessel. Thus, supra-arterial
myotomy for myocardial bridging and milking effect of the LAD
was reported by several surgical centers. However, this approach
is more invasive than that of a catheter-based intervention
and probably also carries a higher risk of postprocedural complications.
In addition, the unpredictable intramural course of the coronary
artery may require deep incision of the ventricular wall, potentially
leading to subsequent ventricular wall aneurysm. Alternatively,
internal mammary artery anastomosis to the LAD may be the treatment
of choice in patients with unsuccessful coronary stenting or
in-stent restenosis. Likewise, patients with significant coronary
disease in other vessels who require coronary bypass surgery
might be suitable candidates for internal mammary artery grafting.
Conclusions.
Recent techniques available in the cardiac
catheterization laboratory, such as QCA, IVUS, and intracoronary
Doppler, have shown that significant myocardial bridging of
a coronary artery, especially the LAD, is characterized by the
following morphological and hemodynamic alterations:
1) phasic systolic vessel compression;
2) persistent diastolic lumen diameter reduction;
3) increased blood flow velocities;
4) retrograde systolic flow; and
5) reduced CFR.
These alterations, particularly in association with aggravating
factors such as increased heart rate, decreased systolic blood
pressure, and coronary vasospasm, may explain the occurrence
of symptoms and myocardial ischemia in patients with symptomatic
MB. As a rule, these patients present with symptoms and signs
of ischemia, as well as a prior history of unstable angina and
non-Q-wave AMI in some patients. In this respect, they are not
strikingly different from patients with significant single vessel
coronary disease. The severity of coronary vessel narrowing
during both systole and diastole can be established by QCA and
IVUS and the degree of flow velocity acceleration by intracoronary
Doppler. In addition, highly specific morphological and flow
velocity patterns include the "half moon" phenomenon
on IVUS and the "finger tip" phenomenon on intracoronary
Doppler.High-frequency intraoperative echocardiography has been
used to image the intramyocardial coronary artery before (Fig.
212-10)and after surgical release. Finally, the treatment
options include medical therapy in the majority of patients
and stent implantation, surgical myotomy, or internal mammary
artery bypass grafting in patients with persistent symptoms
despite medical therapy. The long term outcome of such interventions,
however, is not well known and requires further study.
Bourassa, M.G.,MD and others, Symptomatic Myocardial
Bridges: Overview of Ischemic Mechanisms and Current Diagnostic
and Treatment Strategies, JACC,Vol.41,No.3,2003,pp.351-358.
FIGURE
212-1 (Left) Diagram showing
tunneled left anterior descending coronary artery (LAD) (arrowheads).
(Right) Opened left ventricle showing intramyocardial segment.
(Below) Transverse section of LV wall showing tunneled coronary
artery surrounded by myocardium.
FIGURE
212-2 Diagram showing segments
of tunneled and nontunneled epicardial coronary artery with
changes during ventricular systole and diastole. Ao, aorta;
LV, left ventricle; RV, right ventricle.
FIGURE
212-3 Tunneled epicardial coronary
arteries. Two examples of tunneled left anterior descending
coronary arteries. Each artery is surrounded by myocardium.
FIGURE
212-4 Transverse section of
ventricular myocardium showing the "arcade" of tunneled
epicardial coronary arteries (arrows). A, anterior LV, left
ventricle; RV, right ventricle; P, posterior.
FIGURE
212-5 Tunneled epicardial coronary
artery. A. Coronary angiogram showing tunneled segment of epicardial
coronary artery. B. Corresponding segment of tunneled Left circumflex
coronary artery (arrow).
FIGURE
212-6 Tunneled left anterior
epicardial coronary arteries trom two newborn intants. (Left)
Tunneled left anterior descending. (Right) Tunneled marginal
branch of right coronary artery.
FIGURE
212-7 Diagram showing some of
the clinical and anatomic factors in a tunneled epicardial artery
FIGURE
212-8 Diagram showing morphologic
variations in tunneling (length of tunneled segment, depth of
tunneled segment
FIGURE
212-9 Diagram showing extremes
of tunneled coronary arteries: left main (LM) tunneled through
the ventricular septum, total Length of the left anterior descending
(LAD) located within the myocardium, tunneled segment of LAD
becoming intracavitary. AV, aortic valve; LAD, left anterior
descending IC, Left circumflex; LM, Left main; LV, Left ventricular;
PT, pulmonary trunk; PV, pulmonary valve; RVOFT, right ventricular
outflow tract; RV, right ventricle; TV, tricuspid valve.
Waller,B.F.,Hurst's The Heart,10th edition,
Myocardial Bridges ,chp.39,pg.1168.
Figure
212-10 Intravascular ultrasound
imaging of the myocardial bridging in diastole (A) and in systole
(B). A typical half-moon-shaped echolucent area surrounds the
bridge during the entire cardiac cycle (arrows). Note the catheter
artifact in diastole (A) at seven o'clock.
Boorassa,M.G. and others,Symptomatic
Myocardial Bridges:An Overview.JACC,Vol. 41,No.3, Feb.5.2003,
pp.351-359.
Figure
212-11 Coronary angiogram of
a patient with myocardial bridging of the left anterior descending
coronary artery in the right anterior oblique position. (A)
An absence of constriction during diastole is shown. (B) This
depicts the "milking effect" during systole (arrow).
Symptomatic Myocardial Bridges:An
Overview.JACC,Vol. 41,No.3, Feb.5.2003, pp.351-359.
Table 1a. Quantitative
Coronary Angiography in Patients With Symptomatic Myocardial
Bridges
| |
Schwarz
et al. (12)
n=15 |
Schwarz
et al. (13)
n=42 |
Klues
et al. (14)
n=12 |
Haager
et al. (16)
n=11 |
| Systolic
MLD (mm) |
|
|
|
|
| Proximal |
2.7±
0.5 |
3.1 ±
1.3 |
2.8±0.3 |
2.3±0.5 |
| Distal |
2.2
± 0.4 |
2.5
± 0.8 |
2.2±0.3 |
1.8±0.4 |
| Within
MB |
0.7 ±
0.3 |
0.8 ±
0.6 |
0.5±0.2 |
0.6±0.3 |
| % Diameter reduction |
|
|
|
|
|
Mean (SD)
|
83 ±
9 |
71±16 |
81±9 |
74±12 |
| Range |
71-99 |
- |
- |
55-89 |
Mid-diastolic MLD (mm) |
|
|
|
|
|
Proximal |
2.6±0.5 |
3.0±1.0 |
2.9 ±
0.5 |
- |
| Distal |
2.4 ±
0.4 |
2.5±0.8 |
2.2 ±
0.3 |
- |
| Within
MB |
1.7 ±
0.3 |
1.7±0.4 |
1.6±0.4 |
1.4±0.4 |
| % Diameter reduction |
|
|
|
|
| Mean
(SD) |
41 ±
11 |
35 ±
13 |
35 ±
11 |
34 ±
10 |
| Range |
24-58 |
18-69 |
- |
22-58 |
| Length of MB (mm) |
|
|
|
|
| Systolic |
|
|
|
|
| Mean
(SD) |
27 ± 8 |
23
± 8 |
28
± 4 |
25
± 2 |
| Range
|
19-44 |
11-44 |
- |
13-36 |
| Diastolic |
|
|
|
|
| Mean
(SD) |
25 ±
5 |
22 ±
7 |
27 ±
5 |
- |
| Range |
21-42 |
- |
- |
- |
MB = myocardial bridge; MLD = mean lumen
diameter, SD = standard deviation.
Myocardiobridge-Table
1b. lntracoronary Doppler Flow Velocity Measurements at Rest
in Patients With Symptomatic Myocardial Bridges
| |
Schwarz
et al. (12)
n=15 |
Schwarz
et al. (13)
n=42 |
Klues
et al. (14)
n=12 |
Ge
et al. (15)
n=48 |
Haager
et al. (16)
n=11 |
| APV
(cm/s) |
|
|
|
|
|
| Proximal |
21.2 ± 7.4 |
18.3 ± 5.3 |
13.5±4.9 |
- |
- |
| Distal |
14.3 ± 3.2 |
15.6 ± 3.7 |
12.2±4.9 |
|
|
| Within
MB |
26.7 ± 9.6 |
30.6 ± 16.0 |
27.0 ± 13.6 |
19.0 + 7.9 |
35.0 4.0 |
ASPV (cm/s) |
|
|
|
|
|
| Proximal |
12.3 ± 4.6 |
10.2 ± 4.3 |
6.1 ± 3.7 |
|
|
| Distal |
9.1±2.3 |
9.6 ± 3.0 |
6.5±3.3 |
|
|
| Within
MB |
13.7±6.6 |
14.0 ± 5.2 |
14.7 ± 9.8 |
|
|
| ADPV
(cm/s) |
|
|
|
|
|
| Proximal |
25.5 ± 13.3 |
22.4 ± 7.7 |
17.3 ± 5.7 |
|
|
| Distal |
16.5±3.6 |
18.6±4.6 |
15.2 ± 6.3 |
|
|
| Within
MB |
33.1 ± 13.4 |
38.6 ± 19.0 |
31.5 ± 14.3 |
|
|
| MPV (cm/s) |
|
|
|
|
|
| Proximal |
31.5 ± 13.5 |
33.5 ± 9.6 |
28.6 ± 8.8 |
|
|
| Distal |
24.3 ± 11.6 |
29.3 ± 7.8 |
24.7 ± 14.4 |
|
|
| Within
MB |
78.1 ± 21.1 |
70.5 ± 33.7 |
63.7 ± 26.2 |
43.2 ± 18.7 |
|
| DSVR |
|
|
|
|
|
| Proximal |
- |
2.2 ± 0.6 |
- |
- |
- |
| Distal |
- |
2.0 ± 0.5 |
|
|
|
| Within
MB |
2.4 ± 1.0 |
2.5±1.2 |
|
2.9±1.4 |
|
| CFR |
|
|
|
|
|
| Proximal |
2.7 ± 0.8 |
2.9 ± 0.9 |
|
|
|
| Distal |
2.0 ± 0.6 |
2.3±0.05 |
2.5±0.5 |
2.03±0.54 |
2.6±-0.5 |
ADPV = average diastolic peak flow velocity;
APV = average peak flow velocity ASPV = average systolic peak
flow velocity, CFR = coronary flow reserve; DSVR = diastolic/systolic
flow velocity ratio; MB = myocardial bridge; MPV = maximal
Symptomatic Myocardial Bridges:An
Overview.JACC,Vol. 41,No.3,Feb.5.2003, pp.351-359.
Myocardiobridge-Table 2.
Intracoronary Doppler Flow Velocity Measurements During Atrail
Pacing in Patients With Symptomatic MB's
| |
Schwarz
et al. (12) n=15 |
Schwarz
et al. (13) n=42 |
| APV (cm/s) |
|
|
| Proximal |
29.1 ± 12.2 |
20.3 ± 6.0 |
| Distal |
20 ± 4.2 |
17.0 ± 4.5 |
| Within
MB |
63.3 ± 21.3 |
46.3 ± 20.0 |
| ASPV (cm/s) |
|
|
| Proximal |
10.9 ± 3.4 |
10.9 ± 3.9 |
| Distal |
9.1 ± 4.0 |
10.0 ± 4.2 |
| Within
MB |
16.2 ± 7.0 |
15.8 ± 7.5 |
| ADPV (cm/s) |
|
|
| Proximal |
29.1 ± 12.2 |
27.1 ± 9.5 |
| Distal
|
20.2 ± 4.2 |
22.7 ± 6.6 |
| Within
MB |
63.3 ± 21.2 |
64.7 ± 25.0 |
| MPV (cm/s) |
|
|
| Proximal |
34.4 ± 8.0 |
39.8 ± 12.3 |
| Distal |
27.8 ± 8.4 |
33.8 ± 8.8 |
| Within
MB |
104.80 ± 35.7 |
104.7± 36.7 |
| DSVR |
|
|
| Proximal
|
|
2.3 ± 0.4 |
| Distal |
|
2.7 ± 1.0 |
| Within
MB |
4.9 ± 2.9 |
4.4 ± 2.8 |
Symptomatic Myocardial Bridges:An Overview.JACC,Vol.
41,No.3,Feb.5.2003,pp.351-359.
Figure
212-12 Intracoronary Doppler
blood flow velocity profile showing the characteristic "finger-tip"-like
flow velocity acceleration during early diastole (single arrow)
followed by a plateau phase at mid-to-late diastole (arrows
in A and B). During systole, there is almost no flow within
the bridged segment, but a retrograde flow phenomenon occurs
at the entry site of the myocardial bridge (arrows in A). During
rapid atrial pacing, absolute diastolic flow velocities are
increased, and the duration of the plateau phase is reduced
owing to shortened diastole (C). Reproduced from Schwarz et
al. (13), with permission.
Symptomatic Myocardial Bridges:An
Overview.JACC,Vol. 41,No.3,Feb.5.2003,pp.351-359.
"Western Suburban Cardiology"
video of Myocardial Bridge
for broadband connection only
When coronary arteries travel below the epicardial
surface, myocardial bridges may develop. During systole (heart
muscle contraction), the segment of artery surrounded by the
myocardium narrows as the muscle contracts. This gives an appearance
of a focal narrowing or stenosis. In diastole, the artery appears
normal without evidence of focal narrowing. Luckily, most coronary
blood flow occurs during diastole when the heart is not actively
contracting.
Most bridges are seen in the LAD distribution
and are generally thought to be benign. They are present in
more than 5% of otherwise normal angiograms in patients who
do not have evidence of ischemia in the LAD distribution. If
ischemia is proven, treatment can be provided with coronary
revascularization. Infarction, while rare, can occur as demonstrated
in the literature.
Clin Cardiol. 1997 Dec; 20(12): 1032-1036.
Myocardial Bridge as a Cause of
Thrombus Formation and Myocardial Infarction in a Young Athlete
M. Agirbasli, M.D., G. S. Martin, M.D.,
J. B. Stout, M.D., H. S. Jennings, III, M.D., J. W. Lea, IV,
M.D., J. H. Dixon, Jr., M.D.
This paper reports on a young male athlete
who had an acute myocardial infarction after strenuous exercise.
Coronary angiography demonstrated thrombus formation within
a prominent myocardial bridge. Thrombus formation within a myocardial
bridge may be a potential mechanism whereby this anatomic curiosity
may cause a tragic clinical event.
Polish Heart Journal, June 1998, Vol
XLVIII, Nr 6
JANUSZ RZEZ'NICZAK, DARIUSZ ANGERER, RYSZARD
KALAWSKI, ROBERT PARUCKI, MAREK S?OMCZYN'SKI, JAROSLAW MAN'CZAK,
TOMASZ SIMINIAK
[Intracoronary stent implantation for treatment
of myocardial ischemia induced by myocardial bridge. A case
report.]
Pracownia Badan' Serca i Naczyn, Oddzial
Kardiochirurgii Szpitala im. J. Strusia w Poznaniu
Intracoronary stent implantation for
treatment of myocardial ischemia induced by myocardial bridge.
A case report.
A case of 69-year-old male, referred to coronary angiography
due to episodes of unstable angina is presented. Angiography
has shown a myocardial bridge over the left anterior descending
coronary artery, causing systolic lumen reduction of 87%, and
lack of atherosclerotic changes. Myocardial ischaemia produced
by the myocardial bridge was confirmed by perfusion scintigraphy.
Despite intensive pharmacotherapy severe symptoms were present.
Coronary angioplasty followed by intracoronary stent implantation
was performed. which led to decrease in systolic lumen reduction.
In a 3 month follow-up the patient was free of anginal symptoms,
no signs of restenosis were seen on angiography and no myocardial
ischaemia was detected in perfusion scintigraphy.
Augmentation of Vessel Squeezing
at Coronary-Myocardial Bridge by Nitroglycerin: Study by Quantitative
Coronary Angiography and Intravascular Ultrasound
from American Heart Journal
Yoichiro Hongo, MD, Hiroshi Tada, MD, Kenichi
Ito, MD, Yoshio Yasumura, MD, Kunio Miyatake, MD, Masakazu Yamagishi,
MD, Cardiology Division of Medicine, National Cardiovascular
Center, Osaka, Japan.
Abstract and Introduction
Abstract
Background: Nitroglycerin is known to
augment vessel wall squeezing at the site with coronary-myocardial
bridging (CMB). This study was designed to define the mechanism
of nitroglycerin-induced augmentation of CMB in clinical settings.
Methods: We analyzed nitroglycerin reactivity at the site with
CMB in 39 patients. Maximal and minimal diameters of CMB during
a cardiac cycle were measured by quantitative angiography before
and after intracoronary administration of 250 µg nitroglycerin.
In 15 patients, CMB sites were observed by intravascular ultrasound
to determine the intimal thickness and the time-serial change
in vessel area.
Results: Before nitroglycerin, CMB was demonstrated with angiography
in 25 patients, and the remaining 14 patients showed CMB after
nitroglycerin. The maximal diameter during diastole increased
from 1.4 ± 0.4 mm to 1.9 ± 0.4 mm after nitroglycerin,
whereas the minimal diameter during systole decreased from 1.0
± 0.4 mm to 0.7 ± 0.4 mm (P < .01). Thus nitroglycerin
augmented the percent vessel narrowing during systole from 24%
± 21% to 65% ± 16% (P < .01). Under these conditions,
intravascular ultrasound showed the reduction of the cross-sectional
area of the sites with CMB by -38% ± 16% (P < .01)
during systole, and this phenomenon continued to early diastole
(-30% ± 16%). The intimal thickness was 0.32 ±
0.10 mm, which suggests the absence of atherosclerotic disease
at CMB sites.
Conclusions: These results indicate that nitroglycerin-induced
augmentation of the percent narrowing of CMB can be derived
from further systolic compression of the vessel lumen as well
as diastolic expansion, probably because of the increase in
vessel compliance after nitroglycerin. We suggest that the delayed
dilation of coronary lumen during the early diastole may contribute
to the occurrence of myocardial ischemia.
Augmentation of Vessel Squeezing at
Coronary-Myocardial Bridge
from American Heart Journal
Results
Before nitroglycerin, systolic blood
pressure and diastolic blood pressure were 132 ± 17 and
72 ± 7 mm Hg, respectively, and were 116 ± 5 mm
Hg (P < .01) and 66 ± 9 mm Hg (P < .01) after nitroglycerin.
Heart rate slightly increased from 68 ± 13 to 73 ±
15 beats/min (P < .01) after nitroglycerin.
Angiographic Presence of CMB
Of 39 patients examined, CMB was demonstrated
in 25 patients before nitroglycerin ( Figures 1
and 2
). The remaining 14 patients did not show the apparent systolic
coronary narrowing before nitroglycerin ( Figure
3 ). The lumen diameter of CMB sites was 1.4 ± 0.4
mm in diastole and 1.0 ± 0.4 mm in systole (P < .05).
The calculated percent CMB narrowing before nitroglycerin was
24% ± 21% ( Figure
4 ).
The administration of nitroglycerin augmented
the angiographic appearance of CMB ( Figures 1,
2
and 3
), and all the patients including 14 patients who did not show
CMB before nitroglycerin exhibited CMB. Under these conditions,
the lumen diameter of the CMB site was 1.9 ± 0.4 mm in
diastole and 0.7 ± 0.4 mm in systole (P < .01). The
calculated percent CMB narrowing was 65% ± 16% (P <
.01, Figure
4 ).
Morphology of CMB Observed by Ultrasound
In 15 coronary sites in which CMB was
observed by intravascular ultrasound, the maximal cross-sectional
area was 6.2 ± 1.5 mm2 at end diastole. At end systole,
the lumen was narrowed to 3.8 ± 1.3 mm2 or by -38% ±
16% (P < .01). It was interesting that the reduction of the
cross-sectional area continued to the phase of isovolumic relaxation
(-30% ± 16%) when the coronary blood flow velocity was
maximally accelerated ( Figure
5 ).
The thickness of the vessel wall at the sites
with CMB was 0.32 ± 0.10 mm, and this indicated the absence
of atherosclerotic disease at the sites of the CMB.
Interobserver and Intraobserver Variabilities
For measurement of the lumen diameter
by angiography, interobserver correlation coefficient and error
were 0.96 and 5.3% ± 3.8%, and intraobserver correlation
coefficient and error were 0.98 and 3.0% ± 3.3%. As for
the lumen cross-sectional area by intravascular ultrasound,
interobserver correlation coefficient and error were 0.96 and
5.6% ± 3.3%, and intraobserver correlation coefficient
and error were 0.98 and 3.6% ± 3.2%.
Discussion
Previous Studies
CMB was reported to be present in 23% of
the cases examined by pathology[15] and in 0.5% to 1.5% by angiography.[1,2]
Although the functional significance of CMB in myocardial ischemia
is still controversial, there exists the clinical evidence for
the role of CMB in provoking angina or acute myocardial infarction.[3-7]
Therefore demonstration of CMB is clinically important for the
determination of the cause of myocardial ischemia, particularly
in patients without organic coronary stenosis.
Nitroglycerin is known to augment CMB and
to increase the sensitivity to detect CMB by angiography.[12,13]
According to the preliminary data by Erbel et al, [13] nitroglycerin
augmented the systolic narrowing of the CMB by 29%, yielding
the increase in the sensitivity for detection of the CMB. We
also found that the intracoronary injection of nitroglycerin
resulted in increases in percent narrowing of the CMB from 24%
to 65%.
Advantage of the Study
In this study, we quantitatively evaluated
the changes in coronary diameter at the sites of the CMB before
and after nitroglycerin. We found a marked increase in percent
narrowing of the CMB after nitroglycerin. The angiographic augmentation
of the percent narrowing in systole at the sites of CMB was
derived from not only increase in diastolic diameter but also
decrease in systolic diameter. The increase in vessel wall compliance
after nitroglycerin may contribute to the further compression
of the vessel during systole and to an increase in the angiographic
severity of systolic narrowing. Other factors such as change
in myocardial contractility by nitroglycerin on the magnitude
of systolic compression also should be considered.[16,17] However,
we did not use any cardiotonic drugs during the angiographic
procedure, although there remained the possibility of nitroglycerin-induced
increase in cardiac contractility.[17]
By intravascular ultrasound, vessel narrowing
at the cites with the CMB continued to the early diastole. Ge
et al[18] have reported that there was a persistent diastolic
narrowing of 41% that was caused by a delayed diastolic relaxation
within the bridged segment. Krawczyk et al[19] and May et al[20]
suggested that the delayed opening of the coronary vessel after
systolic compression by CMB could contribute to the genesis
of myocardial ischemia through the insufficient inflow time
and decreased coronary perfusion pressure distal to the CMB.
Under these conditions, measurement of coronary flow velocity
by Doppler flow wire at CMB sites[18,19] before and after nitroglycerin
might be helpful to gain an insight into to the physiologic
significance of nitroglycerin-induced augmentation of CMB.
It is also interesting to point out that the
vessel wall of the CMB was free from atherosclerotic disease.
Continuous changes in vessel diameter may increase local shear
stress,[22] contributing to preventing the occurrence of disease
in the CMB segments, although the exact mechanism of this phenomenon
is still unclear.
Clinical Implications and Limitations
In 14 of 39 patients, coronary angiography
could not demonstrate CMB without the administration of nitroglycerin.
This suggests the role of nitroglycerin in the improvement of
sensitivity of the angiographic detection of the CMB. In fact,
the previous study indicated that the CMB could be demonstrated
by angiography in <10% of the patients examined, although
pathologic examination demonstrated the presence of CMB in >20%
cases studied. This may be important for the selection of the
treatment because medical therapy such as a ß-blocking
agents may be the first choice.[21,23] One might speculate that
the evaluation of the CMB should be done after the appropriate
dose of nitroglycerin.
By the observation with ultrasound, systolic
narrowing of the CMB remained until the early diastole. This
may be related to the fact that tachycardia that mainly shortens
the diastolic period is primarily important in typical myocardial
ischemia associated with CMB.[19,20] From this point of view,
the ß-blocking agent is the most favorable drug to eliminate
the occurrence of myocardial ischemia, although another mechanism
such as reduction of myocardial contractility by ß-blockers
may also be important to reduce the CMB. It is also interesting
to consider whether the severity of CMB after nitroglycerin
is related to the occurrence of myocardial ischemia. Further
study with myocardial perfusion testing may demonstrate the
significance of nitroglycerin-induced augmentation of CMB in
determining the severity of myocardial ischemia in the clinical
setting.
There remains a limitation to this study.
The accuracy of quantitation of CMB by the single-plane dimension
from angiography is limited. Actually, the mean degree of the
reduction in systolic lumen diameter determined by quantitative
angiography was less severe than that reported in previous studies.[2]
Also, there was a difference between the cross-sectional area
variation derived from intravascular ultrasound and angiography
during cardiac cycles. This may be explained by the fact that
the analysis of ultrasound measurements did not include the
cases in whom the ultrasound transducer could not be passed
through the CMB because the lumen diameter was too small.
Conclusions
The present data indicate that intracoronary
nitroglycerin can improve sensitivity for the angiographic detection
of CMB, where the presence of atherosclerotic disease is infrequent.
This effect of nitroglycerin appears to be derived from further
systolic compression of the vessel lumen as well as diastolic
relaxation probably caused by the increase in vessel wall compliance.
We suggest that the delayed dilation of coronary lumen during
early diastole may contribute to the occurrence of myocardial
ischemia.
High-Resolution Imaging of Myocardial Bridging
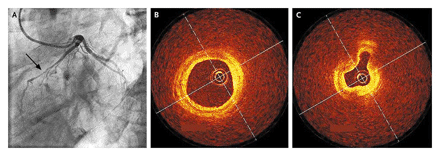
Panel A shows by arrow the results of coronary angiography demonstrating a myocardial bridge, which decreases the diameter of the left anterior descending coronary artery during systole. Optical coherence tomography showed no evidence of atherosclerosis. Instead it did show vessel patency during diastole (Panel B) and collapse during systole (Panel C).
The above images are from the New England Journal of Medicine 358;4, page 392, January 24, 2008 by Dirk Bose,M.D. and Sebastian Philipp, M.D.