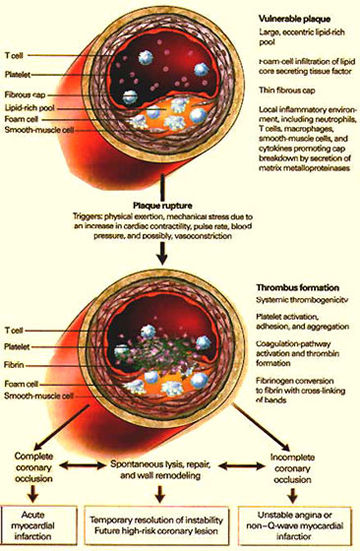
Figure 70
Pathophysiologic Events Culminating in the Clinical Syndrome of Unstable Angina.
Numerous physiologic triggers probably initiate the rupture of a vulnerable plaque. Rupture leads to the activation, adhesion, and aggregation of platelets and the activation of the clotting cascade, resulting in the formation of an occlusive thrombus (clot). If this process leads to complete occlusion of the artery, then acute myocardial infarction with EKG ST-segment elevation (which indicates heart muscle injury) occurs. Alternatively, if the process leads to severe stenosis but the artery nonetheless remains patent, then unstable angina occurs.
Yeghiazarians, Y., Braunstein, J.B., Askari, A., Stone, P.H., Medical Progress: Unstable Angina Pectoris, The New England Journal of Medicine, Vol. 342, Jan 13-00, p 101-114. (modified)
As this lipid like material accumulates, it bulges into the opening of the arteries, leading to varying degrees of obstruction of the blood flow to the heart muscle.
As the obstruction increases there is less reserve for the heart to do work because of the reduction of blood and oxygen to the heart muscle (see figure 53).
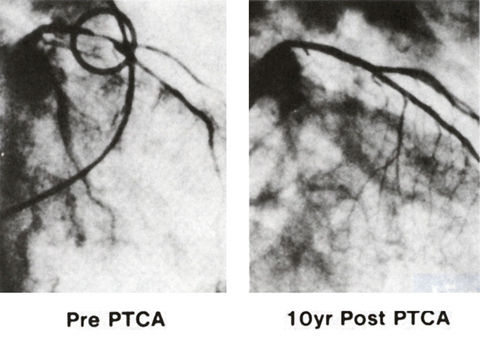
Figure 53
Right anterior oblique coronary arteriogram of the first patient who underwent the first transluminal coronary angioplasty on September 16, 1977 (left) by Andreas R. Gruentzig, M.D., and on September 16, 1987 (right). During this 10-year period, the patient remained completely asymptomatic and the arteriogram at 10 years showed no narrowing in the coronary arteries.
Douglas, Jr., J.S., M.D., King, III, S.B., M.D., Techniques of Percutaneous Transluminal Angiopasty and Atherectomy of the Coronary Arteries, The Hurst's The Heart, 8th ed., p 1346. .
Ultimately there may be enough obstruction to cause chest pain (stable angina pectoris) with exertion due to a lack of oxygen.
These fatty plaques with physical exertion, mechanical stress and other factors like systemic inflammation (manifested by elevated C-reactive protein and amyloid proteins) can rupture into the lumen (opening) of a coronary artery with subsequent blood clot formation (thrombosis), and cause complete obstruction, and hence a heart attack (myocardial infarction), or partial blockage with unstable angina pectoris and rest chest pain (see figures 66 and 67).
There may be simultaneous, multiple,
complex plaques in various coronary arteries, which may become
unstable, making the prognosis more guarded and treatment more
difficult (figure 56d),
due to recurrent ischemia from more generalized pathophysiologic
processes with the potential to destabilize these atherosclerotic
plaques.
Such patients are more likely to require early coronary artery
bypass surgery or staged multivessel angioplasty.
With unstable angina pectoris, the exercise threshold for inducing
pain is markedly reduced and of longer duration.
The resulting reduced blood flow caused by the blood clot accounts
for the rest pain and the reduced threshold for pain induction.
The difference between an atherosclerotic plaque causing either
unstable angina or myocardial infarction is whether or not the
clot is occlusive for a long enough time to cause tissue death
downstream.
Vasoconstriction (spasm) is often enhanced in diseased (atherosclerotic)
coronary arteries (see figure 70).
Treatment
and prevention of atherosclerosis (i.e. coronary arteries) involves
the following recommendations:
1) Periodic
physical examination with laboratory assessment of cholesterol,
triglycerides, blood sugar, thyroid function tests amongst various
tests including EKG.
The cholesterol is fractioned into both low density lipoproteins(LDL) and high density lipoproteins(HDL).
If the cholesterol and especially the LDL are elevated, there are various medications, which help to lower these lipids.
Nicotinic acid(niacin), clofibrate, lovastatin, pravastatin, lipitor(atorvastatin) are some of these medicines.
The atorvastatin also lowers the triglycerides and increases HDL.
The statins inhibit an enzyme called 3-hydroxy-3-methylglutaryl-coenzyme (hmg-coA) reductase, which is involved in the biosynthesis of cholesterol.
The statins have been shown to not only reduce the progression of coronary atherosclerosis, but also to cause regression of the lipid plaques.
But statins have beneficial effects beyond their cholesterol lowering capacity.
They improve endothelial function by reducing oxidative stress within the vascular wall.
The statins also have direct effects on vascular biology, such as activation of the atheroprotective enzyme nitric oxidase synthase, and probably they influence the inhibitory effect on the HMG-CO-A reductase pathway.
Thus, in addition to reducing oxidative stress, statins counteract the inflammatory process in atherosclerotic plaques, and interfere with vascular wall proliferative processes (see figure 70).
Accordingly, besides reducing the lipid pool within the plaque, cholesterol lowering with statins may stablize the atherosclerotic plaque and prevent cardiovascular events.
Hypercholesterolemia is also associated with hypercoaguability and enhanced platelet reactivity at sites of acute vascular damage.
There is accumulating evidence that statins may favorably affect thrombus formation, red cell deformatoin and levels of plasminogen activator inhibitor-1 and fibrinogen, suggesting that statin therapy improves hemorheologic characteristics early in the course of therapy.
Thus, both lovastatin and pravastatin significantly reduce frequency and intensity of ischemic episodes detected by Holter monitoring after as little as 16 weeks of therapy.
Recently, it has been found that lipid lowering drugs are associated with a reduction of reoccurence of ventricular arrhythmias (VA'S) after ICD implantation in patients with coronary artery disease (CAD) and life threathing VA'S.
Also, a recent study has shown that the statin therapy may be effective in the primary prevention of coronary events among persons with relatively low lipid levels but with elevated levels of C-reactive proteins. These patients with elevated levels of C-reactive proteins, even in the absence of hyperlipidemia, are associated with an increased risk of coronary events. Statin therapy reduces the level of C-reactive protein levels independently of its effect on lipid levels. Half of all heart attacks occur among persons without overt hyperlipidemia. Figure 176 shows two views of the role of Acute-Phase Response in Atherothrombosis.
Reference:Ridker,P.M. and Others,Measurement of C-Reactive Protein for the Targeting of Statin Thereapy in the Primary Prevention of Acute Coronary Events,N.Engl.Journal Med.,Vol.344,June28,2001,Pp.1959-1965.
Refernce:Munford,R.S.,Statins and the Acute-Phase Response,N.Engl.Journal Med.,Vol.344,June28,2001,Pp.2016-2017.
ACC/AHA/NHLBI CLINICAL ADVISORY ON STATINS
PREAMBLE
The voluntary withdrawal of cerivastatin (Baycol) from the U.S. market on August 8, 2001, by the manufacturer, in agreement with the Food and Drug Administration (FDA), has prompted concern on the part of physicians and patients regarding the safety of the cholesterol-lowering class of drugs called HMG CoA reductase inhibitors, more commonly known as "statins."
INTRODUCTION
In the literature, the general
terminology used to describe muscle toxicity is inconsistent.
Therefore, for the purpose of this document, the following terms
are used as defined here:
Myopathy-a general term referring to any disease of muscles;
myopathies can be acquired or inherited and can occur at birth
or later in life (Source: NINDS Myopathy Pagehttp://accessible.
ninds.nih.gov/health_and_medical/ disorders/myopathy.htm). Myalgia-muscle
ache or weakness without creatine kinase (CK) elevation. Myositis-muscle
symptoms with increased CK levels. Rhabdomyolysis- muscle symptoms
with marked CK elevation (typically substantially greater than
10 times the upper limit of normal LNII) and with creatinine
elevation (usually with brown urine and urinary myoglobin).
Statins are powerful low-density lipoprotein (LDL)lowering drugs
that are widely used in clinical practice. Results from clinical
trials with a mean duration of 5.4 years have demonstrated a
decrease in CHD and total mortality, reductions in myocardial
infarctions, revascularization procedures, stroke, and peripheral
vascular disease . These trials documented a benefit in both
men and women, primarily in middle-aged and older persons treated
in the setting of either primary or secondary prevention. More
than 50,0 individuals have been randomized to either a increase
in mortality was observed in the drug treatment groups. These
agents reduce the risk of essentially every clinical manifestation
of the atherosclerotic process; they are easy to administer,
with good patient acceptance. There are very few drug to drug
interactions. Although the experience with the safety of statin
therapy outside of clinical trials has not been filly reported,
it is reasonable to suspect that the incidence of side effects
may be higher in clinical situations where patients are not
monitored as closely as they are in clinical trials .
The NCEP has published updated guidelines for treatment of high
blood cholesterol (Adult Treatment Panel III report). These
guidelines are endorsed by the ACC and AHA. They identify elevated
LDL cholesterol as the primary target of therapy and establish
goals for LDL cholesterol that depend on a patient's risk status.
The Adult Treatment Panel III report was able to apply rigorous
clinical trial evidence to identify additional high-risk individuals
for treatment, greatly expanding the number of patients who
are candidates for these drugs. These include patients with
established CHD, other forms of atherosclerotic disease, diabetes
mellitus, multiple risk factors imparting high risk, and severe
hypercholesterolemia. In many patients, relatively high doses
of statins will be required to achieve LDL cholesterol goals
of therapy. In addition, for patients with high triglycerides,
non-high-density lipoprotein (HDL) cholesterol (LDL + VLDL (very
low density lipoprotein) cholesterol) has been identified as
a secondary target of therapy. To achieve the non-HDL cholesterol
goal, many patients will require statin therapy as well. This
broad expansion of statin use will require that increased attention
be given to every aspect of statin therapy (i.e., efficacy,
safety, and cost-effectiveness).
In view of the demonstrated safety of these agents, both medical
professionals and the public were surprised by the recent withdrawal
of a relatively new statin, cerivastatin (Baycol), from the
market. Cerivastatin was first approved for use in the U.S.
in 1997. In August 2001, the manufacturer, Bayer AG, announced
the withdrawal of all dosages of its cholesterol-lowering drug
with the brand names BaycollLipobay (cerivastatin) because of
increasingly frequent reports of serious myopathy, including
severe and life-threatening rhabdomyolysis. Rhabdomyolysis was
reported most frequently when cerivastatin was used at higher
doses and, particularly, in combination with another lipid-lowering
drug, gemfibrozil (LOPID and generics). At the time of withdrawal,
the FDA had received reports of 31 U.S. deaths due to severe
rhabdomyolysis associated with the use of cerivastatin, 12 of
which involved concomitant gemfibrozil use (http://www.fda.gov/cder/drug/infopage/
baycol!). Subsequently, the Wall Street Journal (1/21/02, pg.
AlO) reported that Bayer AG had indicated that as many as 100
deaths have been linked to Baycol. The FDA reports that the
rate of fatal rhabdomyolysis is 16 to 80 times more frequent
for cerivastatin as compared to any other statin .
INCIDENCE OF ADVERSE EVENTS
The statins are well tolerated
by most persons. Elevated hepatic transaminases generally occur
in 0.5% to 2.0% of cases and are dose-dependent . Whether transaminase
elevation with statin therapy constitutes true hepatotoxicity
has not been determined. Progression to liver failure specifically
due to statins is exceedingly rare if it ever occurs . Reversal
of transaminase elevation is frequently noted with a reduction
in dose, and elevations do not often recur with either re-challenge
or selection of another statin . Cholestasis and active liver
disease are listed as contraindications to statin use; however,
no specific evidence exists showing exacerbation of liver disease
by statins. Furthermore, statins have not been shown to worsen
the outcome in persons with chronic transaminase elevations
due to hepatitis B or C, and treatment of hyperlipidemia may
actually improve transaminase elevations in individuals with
fatty liver . An observational study has suggested a rare association
of statin use with polyneuropathy. This has not been found in
the large blinded randomized controlled trials.
The ability of statins to produce myopathy under some circumstances
is well established. A common complaint is non-specific muscle
aches or joint pains that are generally not associated with
significant increases in creatine kinase. In placebo-controlled
trials, the incidence of these complaints (generally reported
as about 5%) is similar between placebo and active drug therapy,
suggesting they may not be drug-related . Nonetheless, in some
patients, the temporal association with statin therapy is strong
enough to implicate these drugs as a cause of these complaints.
Other patients can have mild-to-moderate elevations of creatine
kinase without muscle complaints. Again, elevations may be non-specific,
but a statin effect often cannot be niled out.
It is rare that patients treated with a statin exhibit severe
myositis characterized by muscle aches, soreness or weakness
and associated with elevated creatine kinase levels, generally
greater than 10 times the ULN. In this setting, failure to discontinue
drug therapy can lead to rhabdomyolysis, myoglobinuria, and
acute renal necrosis . Myositis is most likely to occur in persons
who have complex medical problems and/or who are taking multiple
medications. It may rarely occur with statin monotherapy, but
it occurs more frequently when statins are used in combination
with a variety of medications, including cyclosporine, fibrates,
macrolide antibiotics, certain antifungal drugs, and niacin
. Some of the drug to drug interactions involve specific interactions
with the cytochrome P-450 drug-metabolizing system, especially
those involving the 3A4 isozyme . The combination of statins
with a fibrate is attractive for persons who have both high
serum cholesterol and high triglycerides or for those who continue
to have elevated triglycerides after reaching their LDLcholesterol
target on statin therapy. However, there may be a concern about
an increased danger of developing myopathy with this combination.
In the past, this combination was thought to be "contraindicated"
because of the potential danger of myopathy. More recently,
it has been used increasingly with apparent safety in the majority
of persons. This combination is now presented by the ATP III
report as an option, with careful monitoring, for some forms
of dyslipidemia.
The FDA report comparing the rate of fatal rhabdomyolsis among
different statins is of considerable importance . The FDA performed
a detailed review of all reports of fatal rhabdomyolysis in
their Adverse Event Reporting System and obtained the number
of prescriptions dispensed since marketing of each statin began
in the U.S. Fatal rhabdomyolysis was extremely rare (less than
1 death! million prescriptions). As previously noted, the rate
of fatal rhabdomyolsis for cerivastatin was far greater than
that for other statins (16 to 80 times higher). Even after excluding
cases in which cerivastatin was administered with gemfibrozil,
the reporting rate for fatal rhabdomyolysis with cerivastatin
monotherapy (1.9 deaths per million prescriptions) was 10 to
50 times higher than for other statins. The FDA report also
noted that more than 60% of the fatal cases with cerivastain
were associated with use of the highest dose (0.8 mg daily).
The FDA notes that the data are reporting rates, not incidence
rates. Thus, statistically "rigorous comparisons between
drugs . . . are not recommended" . Nevertheless, review
of these data strongly suggests that there were no clinically
important differences in the rate of fatal complications among
the five statins now available in the U.S. (atorvastatin, fluvastatin,
lovastatin, pravastatin, and simvastatin). Clinicians should
consider the rates of severe myopathy as equivalent among all
of these approved statins.
The following are summary comments reflecting current experience
with these issues:
Statin therapy appears to carry
a small but definite risk of myopathy when used alone. According
to several large clinical trial databases, the incidence of
severe myopathy is reported to be 0.08% with lovastatin and
simvastatin . Elevations of CK greater than 10 times the ULN
have been reported in 0.09% of persons treated with pravastatin.
All currently marketed statins appear to have a similar potential
for causing this adverse effect.
Fibrate treatment alone appears to be associated with some (probably
similar) risk of myopathy.
Of the nearly 600 persons who have participated in controlled
clinical trials of a statin and fibrate combination, 1% have
experienced a CK greater than 3 times the ULN without muscle
symptoms, and 1% have been withdrawn from therapy because of
muscle discomfort . None of these findings were considered serious
by the trial investigators. No cases of rhabdomyolysis or myoglobinuria
have been encountered in these clinical trails.The experience
in these trails is predominantly with lovastatin and gemfibrozil,
but it is reasonable to believe that the experiences with other
statin-fibrate combinations would be similar.
MECHANISM OF MYOPATHY
Because it occurs so rarely, little is known about the fundamental mechanisms of stalin-associated myopathy. It has been suggested that statins lead to inhibited synthesis of compounds arising from the synthetic pathway of cholesterol. In theory, this could lead to ubiquinone (an essential intracellular energy component) deficiency in muscle cell mitochondria, disturbing normal cellular respiration and causing adverse effects including rhabdomyolysis. Despite in-vitro support for this concept , a human study of six months of simvastatin treatment (20 mg per day) on skeletal muscle concentrations of high-energy phosphates and ubiquinone demonstrated that the muscle high-energy phosphate and ubiquinone concentrations assayed after simvastatin treatment were similar to those observed at baseline and did not differ from values in control subjects . No clinical study has yet provided support for the hypothesis of diminished isoprenoid synthesis or energy generation in muscle cells during statin therapy. Some have proposed that statin interaction with the cytochrome P-450 hepatic enzyme system might be related to myopathy . Support for this concept comes, in part, from the known enhanced toxicity when statins are administered with agents sharing metabolism by the same cytochrome isoforms. Finally, it has been shown that exercise in combination with lovastatin produces greater creatine kinase elevations than those produced by exercise alone, suggesting that statins can exacerbate exercise-induced skeletal muscle injury.
DIAGNOSIS
Routine laboratory monitoring of CK is of little value in the absence of clinical signs or symptoms. Therefore, all persons beginning to receive statins should be instructed to report muscle discomfort or weakness or brown urine immediately, which should then prompt a CK measurement.
MANAGEMENT
Baseline Measurements
Before initiating statin therapy, baseline measurements, including a lipid and lipoprotein profile, that will be used to follow the drug's efficacy and safety should be documented. Current labeling for all statins requires baseline measurements of liver function, including alanine transferase and aspartate transferase, although this is not agreed on by many liver experts and will likely undergo review in the future. Modest transaminase elevations (less than 3 times the ULN) are not thought to represent a contraindication to initiating. continuing, or advancing statin therapy, as long as patients are carefully monitored,Many experts also favor,and the ATP III report recommends, baseline CK measurement, reasoning that asymptomatic CK elevations are common and pre-treatment knowledge of this condition can aid in later clinical decision making.
Monitoring for Adverse Reactions and Adjusting Therapy
Once therapy has been initiated,
symptoms may appear at any time. If myositis is present or strongly
suspected, the statin should be discontinued immediately. Several
key points should be kept in mind.
Obtain a CK measurement if the patient reports suggestive muscle
symptoms, and compare to CK blood level prior to beginning therapy.
Because hypothyroidism predisposes to myopathy, a thyroid-stimulating
hormone level should also be obtained in any patient with muscle
symptoms.
If the patient experiences muscle soreness, tenderness, or pain,
with or without CK elevations, rule out common causes such as
exercise or strenuous work. Advise moderation in activity for
persons who experience these symptoms during combination therapy.
Discontinue statin therapy (or statin and niacin or fibrate
if the patient is on combination therapy) if a CK greater than
10 times the ULN is encountered in a patient with muscle soreness,
tenderness, or pain.
If the patient experiences muscle soreness, tenderness, or pain
with either no CK elevation or a moderate elevation (3 to 10
times the ULN), follow the patient's symptoms and CK levels
weekly until there is no longer medical concern or symptoms
worsen to the situation described previously (at which point
therapy should be discontinued). For patients who develop muscle
discomfort and/or weakness and who also have progressive elevations
of CK on serial measurements, either a reduction of statin dose
or a temporary discontinuation may be prudent. A decision can
then be made whether or when to reinstitute statin therapy.
Asymptomatic Patients With CK Elevation
Prior to the withdrawal of cerivastatin,
the ATP III report did not recommend routine ongoing monitoring
of CK in asymptomatic patients. If a physician chooses to obtain
CK values in asymptomatic patients, particularly those on combination
therapy, and CKs are elevated to more than 10 times the ULN,
strong consideration should be given to stopping therapy. Following
discontinuation, wait for symptoms to resolve and CK levels
to return to normal before reinitiating therapy with either
drug and use a lower dose of the drug(s) if possible.
Some asymptomatic patients will have moderate (i.e., between
3 and 10 times the ULN) CK elevations at baseline, during treatment,
or after a drug holiday. Such patients can usually be treated
with a statin without harm. However, particularly careftil monitoring
of symptoms and more frequent CK measurements are indicated.
Increased Risk States for Statin-Associated Myopatby
Prevention of statin-associated myopathy can best be accomplished by attention to those factors that might increase the risk for such myopathy:
o Advanced age (especially more than 80 years) in patients (women more than men)
o Small body frame and frailty
o Multisystem disease (e.g., chronic renal insufficiency, especially due to diabetes)
o Multiple medications
o Perioperative periods
o Specific concomitant medications or consumption as listed below (check specific statin package insert for warnings)
Fibrates (especially gemfibrozil, but other fibrates too)
Nicotinic acid (rarely)
Cyclosporine
Azole antiftingals
Itraconazole and ketoconazole
Macrolide antibioticso Erythromycin and clarithromycin
o HIV protease inhibitors
o Nefazodone (antidepressant) Verapamil
Amiodarone
Large quantities of grapefruit juice (usually more than 1 quart per day)
Alcohol abuse (independently predisposes to myopathy)
Clinical Precautions
Most myopathy associated with
statins appears to occur in patients who are at risk for the
condition. For this reason, physicians should be aware of several
caveats when prescribing statin therapy. Myopathy is more likely
to occur at higher statin doses than at lower doses. For this
reason, doses should not exceed those required to attain the
ATP III goal of therapy. As a rule, statin therapy should be
employed more cautiously in older persons, particularly older
thin or frail women, but it is not contraindicated in these
or other high-risk patients. Among older persons, those with
multisystem disease apparently are at higher risk. Patients
with diabetes combined with chronic renal failure also appear
to be at higher risk for myopathy-such patients should be monitored
careftilly. In several instances, myopathy has developed when
patients were continued on statin therapy during hospitalization
for major surgery. Therefore, it probably is prudent to withhold
statins during such periods.
Particular attention should be given to drug interactions when
employing statin therapy. Although the combination of statin
plus fibrate is accompanied by an increased danger of myopathy,
the use of moderate statin doses combined
Table 1. Summaiy of HMG CoA Reductase Inhibitors
| Available drugs | Lovastatin, pravastatin, simvastatin, fluvastatin, atorvastatin |
| Lipid/lipoprotein effects | LDL cholesterol 1 18-55 percent |
| HDL cholesterol 5-15 percent | |
| Triglycerides decreasel 7-30 percent | |
| Major use | To lower LDL-cholesterol |
| Contraindications | |
| -----Absolute | Active or chronic liver disease |
| -----Relative | Concomitant use of cyclosporine, gemfibrozil, or niacin, macrolide |
| antibiotics, various anti-flingal agents, and cytochrome P-450 inhibitors | |
| Efficacy | Reduce risk for CHD and stroke |
| Safety | Side effects minimal in clinical trials |
| Usual starting dose | Lovastatin - 20 mg |
| Pravastatin - 20 mg | |
| Simvastatin - 20 mg | |
| Fluvastatin - 20 mg | |
| Atorvastatin - 10 mg | |
| Maximum FDA-approved Dose |
Lovastatin - 80 mg |
| Pravastatin - 80 mg | |
| Simvastatin 80 mg | |
| Fluvastatin - 80 mg | |
| Atorvastatin - 80 mg | |
| Available preparations | Lovastatin - 10, 20, 40 mg tablets |
| Pravastatin - 10, 20, 40, 80 mg tablets | |
| Simvastatin - 5, 10, 20, 40, 80 mg tablets | |
| Fluvastatin - 20, 40, 80 (xl) mg tablets | |
| Atorvastatin - 10, 20, 40, 80 mg tablets |
with fibrate appears to have a relatively low incidence of myopathy,
especially when used in persons without multi-system disease
or multiple medications. The combination of statin plus nicotinic
acid seemingly carries a lower risk for myopathy than does statin
plus fibrate. Finally, physicians should be aware of the dangers
of interactions of statins with the other drugs previously listed.
These combinations should also be used with caution or avoided
altogether. Furthermore, it is important for clinicians prescribing
statins to make sure that their patients are aware of these
potential drug interactions, because in current practice, a
patient may receive prescriptions from many different care-givers.
Table 2. Monitoring Parameters and Follow-Up
Schedule
| Monitoring Parameters | Follow-Up Schedule |
|
| Statins | Headache, dyspepsia | Evaluate symptoms
initially, 6 to 8 weeks after starting therapy, then at each follow-up visit |
| Muscle soreness, tenderness, or pain |
Evaluate muscle symptoms and CK before starting therapy
Evaluate muscle symptoms 6 to |
|
| ALT, AST | Evaluate ALT/AST
initially, approximately 12 weeks after starting therapy, then annually if indicated |
SUMMARY
Statin therapy holds great promise for reducing the incidence of major coronary events, coronary procedures, and stroke in high-risk patients. At present, this potential has not been fully realized, because many patients at heightened risk are not being treated with these drugs. There is a well documented under-use of statins in clinical practice. Statins have proven to be extremely safe in the vast majority of patients receiving them. Few significant side effects were observed in clinical trials, and post-marketing reports of adverse events have been very limited when considered in comparison to the very large number of persons safely receiving these drugs. Even so, these drugs are not entirely free of side effects, and as for all drugs, they should be used appropriately and judiciously. This advisory encourages the appropriate use of statins while pointing out the possibility of side effects in certain patients. If statins are used with appropriate caution in these selected patients, the likelihood of developing clinically important myopathy should be substantially reduced. (See Tables 1 and 2.
Pasternak ,R. and others,JACC,Vol.40,No.3,2002,pp.567-572.
The fasting blood sugar is aimed at diagnosing diabetes mellitus, which can be associated with hypercholesterolemia. If the sugar is found to be elevated, a 3 hour glucose tolerance test is indicated to establish the diagnosis.
Treating the diabetes can result in a lowering of the cholesterol.
Thyroid function studies are needed to diagnose hypothyroidism associated with hypercholesterolema.
Replacement with thyroid hormone would help lower the cholesterol.
2) Elimination of obesity is very important in the treatment and prevention of atherosclerosis.
Certainly, to lose the extra weight is going to require marked calorie restriction and limitation on whole dairy products, eggs, meat, poultry skin, fried and processed food, especially if the cholesterol is elevated.
If diabetes is present, sugar should be markedly curtailed.
With hypertension, less salt is a must.
Foods to favor include the the following:
Fresh foods, legumes and vegetables, especially deep greens
and yellow-oranges (3-5 servings a day).
Fruits (2-4 servings/day).
Grains, especially whole grains (at least 6servings/day).
Fish (2 or more portoins/week).
Nonfat dairy products.
A dietician's consult may be in order.
3) Regular exercises is desirable;for example,walking for at least 2-4miles a day is recommended. Brisk walking is recommended for fitness and endurance.
4) Cessation of smoking.
Smoking appears to be a major contributor to increased risk of coronary atherosclerosis.
Vascular endothelium can be damaged by cigarette smoking,which can cause thromboxane A-2 release, decrease prostacyclin production,induce alpha-adrenergic stimulation, enchance vasopressin generation, and finally provoke constriction of the vessel itself.
Platelet aggregation is increased and survival altered by cigarette smoking.
It also elevates the levels of catecholamines and free-fatty acids, which can further alter vascular tone, resulting in increased heart rate, blood pressure,and myocardial contractility with associated increased vascular resistance, leading to increased oxygen demand.
Cigarette smoking also is associated with reduced levels of HDL-cholesterol.