Pulmonary
stenosis occurs in 10-12% of cases of congenital heart disease
in adults. The obstruction is vascular in 90% of patients, but
may occur above or below the valve itself. There may be associated
other congenital heart defects. Although the three valve cusps
(see fig 25) in stenosis are usually thin and pliant, their
commissures are fused (see fig 25), leading to a dome-shaped
valve with a small central opening during ventricular contraction
(systole).
The
other 10% of cases have thickened, immobile and myxomatous valves.
The
normal valve area is 2.0 cm2 per square meter of body surface
area, with no pressure gradient across the valve during systole.
When the valve becomes stenotic, the right ventricle systolic
pressure increases, creating a gradient across the valve.
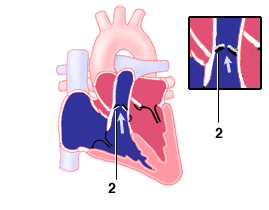
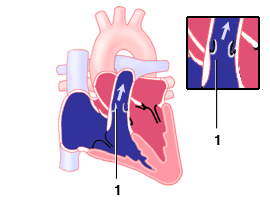
Fig1-1:Normal pulmonary valves with cusps separating widely
during systole;Fig2-2:Pulmonary valve stenosis with valves bulging
backward like a hood due to not opening .
Pulmonary
stenosis is mild, if the valve area is larger than 1.0 cm2 per
square meter and the trans-valvular gradient is 50-80 mmHg,
or the peak RV systolic pressure is less than 75 mmHg.
The
stenosis is moderate if valve area is 0.5-1.0 cm2 per square
meter, trans-valvular gradient is 50-80 mmHg, or right ventricle
systolic pressure is 75-100 mmHg.
It
is severe when the valve area is less than 0.5 cm2, and the
gradient is more than 80 mmHg.
Diagnosis
includes the following:
1)
Physical examination. A thrill may be felt along the left sternal
border. A murmur may be heard along the left sternal border.
The murmur comes from a narrowing of a segment of the pulmonary
artery above the pulmonary valve or the narrowing can be in
one of the pulmonary artery branches(right or left).The murmur
is a harsh noise peaking in the middle of the cycle of the heart
contracting to push blood through the pulmonary artery. The
blood going through a narrowed segment of the pulmonary artery
creates this noise,best heard just to the left of middle line
of the chest, up close to and under the left collar bone(clavicle)
and can also be heard under the left arm and in the back!
2)
EKG show right ventricular wall thickening (RVH).
3)
Echocardiogram show right ventricular wall thickening (RVH)
and paradoxically septal (IVS) motion during systole, and the
site of obstruction. Doppler studies can assess the degree of
stenosis.
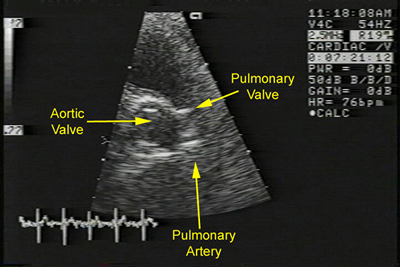
Fig1: Normal echocardiographic parasternal cross sectional
view of pulmonary valve.
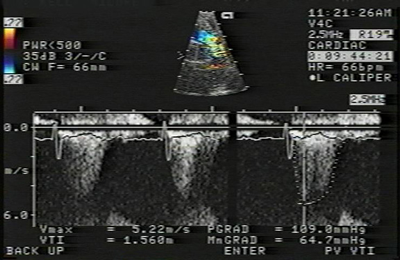
Fig2: Doppler study of case of severe pulmonary stenosis with
a Doppler velocity of 5.22m/sec.(pulmonary gradient of 109mmHg.
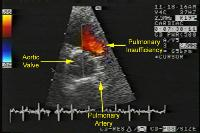
Fig3 Echocardiogram,parasternal ,cross sectional view of a
case of pulmonary stenosis with marked regurgitation
The clinical course of pulmonary stenosis is favorable in most
patients
with mild to moderate obstruction. In a national study, 86%
of patients had no
increase in their pressure gradients over a 4- to 8-year interval.
Those
with a significant increase were less than 4 years of age and
had at least
moderte stenosis initially. Progression during the period of
growth
seems to be the likely explanation for most of the increases,
but a few patients
developed subvalvular muscular hypertrophy, which increased
the
obstruction.
Even mild obstruction may progress significantly in some infants
during
the first year of life. The prognosis of those with severe obstruction
without intervention is poor, especially in infants with critical
obstruction.With severe obstruction,right ventricular damage
and dysfunction can ensure over the years, and heart failure
or arrhythmias can cause premature death in adults.Tricuspid
regurgitation also may result. Obstructon of the subvalvular
type frequently increases with time. Brain abscess, infective
endocarditis may occur.
In the above cited national study reevaluated 15 to 25 years
later, the probability of 25-year survival was 95.6% compared
with an expected age- and sex-matched control group survivalof
96.6%.97% were asymptomatic. Studies suggested no pulmonary
stenosis in 2 %, mild stenosis in 93%, moderate in 3%, and severe
stenosis in only 1%
Treatment depends on the degree of stenosis .Frequent reexaminations
are indicated to detect any evidence of progression,with more
frequent
evaluation for those under one year of age.
Balloon valvuloplasty has replaced surgery therapy as a first
approach (see attached figures).

Fig4:(ps balloon animation pulmonary)Animation of balloon valvotomy
of pulmonary artery with pulmonary stenosis.In this procedure
the cardiologist inserts a special catheter in a large vein
in the groin (femoral vein) and advance it to reach into the
right ventricle and then cross the narrow opening of the pulmonary
valve. With the catheter in place across the pulmonary valve
an elongated balloon over the catheter is inflated, therefore,
stretching the narrow valve opening forcing it to enlarge, thus
relieving the stenosis.The catheter has an inflatable balloon
at its end. Once across the pulmonary valve, the balloon is
transiently inflated causing the valve leaflets to open wide,
thus relieving stenosis.
Balloon valvuloplasty
Long-Term Results of Pulmonary Balloon Valvulotomy in Adult
Patients
Background and aim of the study: The study aim was to define
the long-term outcome of pulmonary balloon valvulotomy (PBV)
in adult patients.
Methods: PBV was performed in 87 patients (46 females, 41 males:
mean age 23± 9 years. range 15-54 years) with congenital
pulmonary valve stenosis (PS). Intermediated follow up catheterization
(mean 14.6± 5.0; range 6- 24 months) was performed after
PBV in 53 patients. Clinical and Doppler echocardiography examinations
were carried out annually in 82 patients (mean 8.0 ±
3.9, range: 2-15 years).
Results: There were no immediate or late deaths. I he mean
catheter peak pulmonary gradient (PO) before and immediately
after PBV, and at intermediate follow-up was 105 t 39, 34 126
(p<0.0001) and 17 t 14 (p<0.0001) butt Hg, respectively.
The corresponding values for right ventricular (RV) pressure
were 125 ± 38. 59 t 21 and 42 t 112 (p<0.0001) turn
Hg respectively. The infundibular gradients immediately after
PBV and at intermediate follow up were 31± 24 and 14
± 9 mm Hg (p<0.0001), whilst cardiac index improved
from 2.68 ± 0.73 to 3. ± 0.4 l/min/m2 (p< 0.05)
at intermediate follow up. Doppler PG before PBV and at intermediate
and long-term follow up were 91 ± 33 ( range 36- 200)
mmHg: 28 ± 12 ( range 10-60) mm Hg (p<0.000I) and
26 ± ImmHg ( p = 0.2), respectively. New pulmonary regurgitation
(PR) was noted in 21 patients (25%) after PBV.
Five patients (6%) with a suboptimal result immediate valve
gradient _=30 mm Hg); developed restenosis and underwent repeat
valvulotomy 6-12 months later using a larger balloon, and with
satisfactory outcome. Moderate to severe tricuspid regurgitation
(TR) in seven patients regressed after PBV.
Conclusion: The long-tern results of PBV in adults are excellent,
with regression of concomitant, severe infundibular stenosis
and/or severe TR. Hence, PBV should be considered as the treatment
of choice for adult patients with PS.
Mohammad E. Fawzy', Mahmoud Awad', Omar Galal', Mohamed Shoukri2,
Hesham Heeg7y', Bruce Dunn', L. Mimish', Zohair Al-1-ialees'
The Journal of Heart Valve Diseases:2001;10.
A thickened,immobile, dysplastc pulmonary valve is best treated
by
complete excision. Sub valvular stenosis is relieved through
a right
vehtriculotomy, a main pulmonary arteriotomy or a right atriotomy.
The blueness of the eyelids may represent cyanosis (desaturation
of the
blood due to venous blood being mixed through a shunt like the
atrial septal
defect with oxygenated blood in the left atrium) and should
be brought
to the cardiologist's attention.
Author: Kurt Pflieger, MD, Consulting Staff, Department of
Pediatrics, Lake Pointe Medical Center http://www.emedicine.com/ped/topic1953.htm
Background: Pulmonary stenosis may be valvar, subvalvar (infundibular),
or supravalvar. These lesions are associated collectively with
obstruction to right ventricular outflow.
Pathophysiology: By far the most common pathology is valvar
pulmonary stenosis, accounting for more than 90% of pulmonary
stenosis. The pulmonary valve may be bicuspid or dysplastic,
as seen in Noonan syndrome.
Isolated infundibular or subvalvar pulmonary stenosis is uncommon
and usually is associated with a ventricular septal defect,
such as in tetralogy of Fallot.
Supravalvar pulmonary stenosis often is associated with rubella
syndrome and Williams syndrome (unusual facies, mental retardation,
hypercalcemia).
Peripheral pulmonary stenosis frequently is observed in newborns
and represents a relative narrowing of the branch pulmonary
arteries occurring as a result of the acute angle of bifurcation
of the main pulmonary artery at this age.
Frequency:
In the US: Frequency of pulmonary stenosis represents 8-12%
of all congenital heart defects. Isolated valvar pulmonary stenosis
with an intact ventricular septum is the second most common
congenital cardiac defect. It may occur in as many as 50% of
all patients with congenital heart disease when associated with
other congenital cardiac lesions.
Mortality/Morbidity: Severity of the valvar dysplasia determines
morbidity and mortality.
Mild-to-moderate valvar pulmonary stenosis is extremely well
tolerated.
Severe pulmonary stenosis can be associated with decreased cardiac
output, right ventricular hypertrophy, early congestive heart
failure (CHF), and cyanosis.
Sex: The male-to-female ratio is equal.
Age: Pulmonary stenosis most commonly presents in infancy.
CLINICAL
History:
Patients who are acyanotic usually are asymptomatic.
In moderate-to-severe cases, the patient may demonstrate exertional
dyspnea and easy fatigability.
Severe cases may present as heart failure and/or cyanosis.
Physical:
Patients usually are acyanotic.
Right ventricular predominance on palpation with or without
a systolic thrill is typical.
A systolic ejection click usually is present at the left upper
sternal border and is variable with respiration, louder on expiration.
Pulmonary component of the second heart sound might be diminished
in intensity.
Systolic ejection murmur (crescendo-decrescendo), grade II-V/VII,
is audible at the left, upper sternal border transmitting into
the back and to the posterior lung field.
The severity of the valvar disease is directly related to the
intensity and duration of the murmur. When severe, the murmur
extends into diastole (beyond the second heart sound).
Hepatosplenomegaly may develop in cases of congestive heart
failure.
Severe valvar pulmonary stenosis associated with tricuspid insufficiency
may be accompanied by elevated central venous pressure, hepatosplenomegaly,
pulsatile liver, jugular venous pulsations, and hepatojugular
reflux.
Peripheral pulmonary stenosis (commonly encountered in the neonate)
usually is associated with a grade II/VI systolic murmur that
radiates into the posterior lung fields and axillae. The pathology
of peripheral pulmonary stenosis is secondary to the acute angular
takeoff of the branch pulmonary arteries from the main pulmonary
arteries specific to a neonate's anatomy. This condition and
the associated murmur usually resolve spontaneously in the first
month of life.
Causes: The development of pulmonary valvar stenosis is primarily
a maldevelopment of the pulmonary valve tissue and distal portion
of the bulbus cordis, which is characterized by fusion of leaflet
commissures, resulting in a thickened and domed appearance to
the valve.
Coexisting cardiac malformations, such as ventriculoseptal
defect (VSD), atrial septal defect (ASD), and patent ductus
arteriosus (PDA), may complicate the anatomy, physiology, and
clinical picture.
Aberrant flow patterns in utero also may be, in part, associated
with maldevelopment of the pulmonary valve.
DIFFERENTIALS
Aortic Stenosis, Valvar
Atrial Septal Defect, Ostium Secundum
Bundle Branch Block, Right
Double Outlet Right Ventricle, Normally Related Great Arteries
Holt-Oram Syndrome
Partial Anomalous Pulmonary Venous Connection
Pulmonary Stenosis, Infundibular
[Tetralogy of Fallot with Pulmonary Atresia]
[Tetralogy of Fallot: Surgical Perspective]
Ventricular Septal Defect, Supracristal
Ventricular Septal Defect: Surgical Perspective
Other Problems to be Considered:
Complex congenital heart disease associated with findings of
pulmonary stenosis
Infundibular/subpulmonary stenosis
Supravalvar pulmonary stenosis
Lab Studies:
Laboratory evaluation usually is not helpful.
Oximetry will provide information of potential right-to-left
shunting in borderline cyanotic lesions or in patients with
anemia but will not identify the cause of the shunt (pulmonary,
interatrial, interventricular, great arterial).
Although arterial blood gas (ABG) analysis usually is not needed,
one notable exception is the hyperoxia test in the newborn with
cyanosis of undetermined origin.
Administered 100% FIO2 generally will not increase the partial
pressure of oxygen to levels much greater than 100 mmHg in patients
with a cyanotic congenital heart defect (right-to-left intracardiac
shunt).
Imaging Studies:
Chest roentgenogram
Demonstrates a prominent main pulmonary artery segment but,
usually, a normal heart size
Pulmonary vascular markings usually are normal but may be decreased
in severe pulmonary stenosis.
Congestive heart failure will present as cardiomegaly with right
ventricular and right atrial enlargement in severe valvar pulmonary
stenosis, with or without tricuspid insufficiency.
Echocardiography
The sine qua non of diagnosis is 2-dimensional and Doppler echocardiography.
A thickened pulmonary valve with restricted systolic motion
(doming) in the parasternal short axis view is demonstrated.
Multiple views will be used to confirm the absence of coexistent
congenital cardiac disease.
Frequently, dilatation of the main pulmonary artery distal to
the stenotic orifice occurs.
Doppler studies will be able to accurately determine the velocity
of flow at single or multiple levels, which then can be converted
to reproducible pressure gradients by means of the modified
Bernoulli equation: pressure gradient (mmHg) = 4 x (velocity
squared [m/s])
Multiple views and measurements will increase the accuracy of
the predicted peak systolic pressure gradient.
Severe pulmonary stenosis with gradients >50 mmHg, as diagnosed
by a continuous wave Doppler recording through the pulmonary
valve, requires balloon valvuloplasty or surgery.
Most children with pulmonary stenosis do not require further
evaluation beyond echocardiography.
Other Tests:
Electrocardiogram
Results usually are normal in mild pulmonary stenosis.
Right axis deviation and right ventricular hypertrophy occur
in moderate valvar pulmonary stenosis.
The degree of right ventricular hypertrophy correlates well
with the severity of pulmonary stenosis.
Right atrial hypertrophy and right ventricular hypertrophy with
strain pattern are observed when pulmonary stenosis is severe.
Superior QRS axis (left axis deviation) is seen with dysplastic
pulmonary valve and Noonan syndrome
Procedures:
Cardiac catheterization
Catheterization is not indicated for mild pulmonary stenosis
but is essential in severe stenosis.
This procedure is used to assess the morphology of the right
ventricle, the pulmonary outflow tract, and the pulmonary arteries.
Patients with echocardiographic evidence of significant pulmonary
stenosis (50-60 mmHg) should undergo diagnostic and therapeutic
cardiac catheterization with preparation for balloon dilatation
of the pulmonary valve.
Angioplasty of a branch of the pulmonary artery stenosis has
been accomplished but carries a significantly higher risk than
valvar pulmonary stenosis.
Infundibular and supravalvar pulmonary stenosis, if severe,
require operative and invasive surgical intervention.
TREATMENT
Medical Care:
Prehospital care: Collect essential information from the vital
signs, including pulse, respiratory rate and work of breathing,
blood pressure (upper and lower extremities), and presence or
absence of cyanosis.
Presence of associated congenital cardiac anomalies should be
anticipated until proven otherwise.
If the patient has a known large left-to-right shunt, such
as PDA or ventriculoseptal defect (VSD) and is in respiratory
distress, diuresis should be attempted and is effective in reducing
the cyanosis secondary to pulmonary edema.
Use of oxygen may reduce pulmonary artery pressure in patients
with a reactive pulmonary vasculature, thereby increasing pulmonary
blood flow.
Administer oxygen in any cyanotic patient with respiratory distress.
Emergency department care
Limited diagnostics are needed after the structural diagnosis
is made.
Frequently, the workup performed for the cyanotic infant with
respiratory distress and hypotension/shock will be that of a
septic patient.
Surgical Care:
Cardiac catheterization with balloon valvuloplasty is the preferred
therapy for severe or critical valvar pulmonary stenosis. In
neonates with critical valvar pulmonary stenosis, balloon dilatation
mortality is lower than surgery mortality and is the treatment
of choice.
Patients are referred for this procedure when the echocardiography
gradient is in the moderate or severe range.
A balloon catheter is placed over a wire in an antegrade fashion
through the femoral vein, inferior vena cava, right atrium,
right ventricle, and across the valve.
The balloon, with diameter 120% of the annulus diameter, is
inflated and deflated while straddling the valve. This usually
results in a significant gradient reduction. Some pulmonary
insufficiency may develop but is well tolerated.
Temporary subvalvar dynamic obstruction may occur and usually
resolves over several days. The procedure usually is well tolerated
but is more risky in infants younger than six months, especially
neonates with critical stenosis.

Fig4:(ps balloon animation pulmonary)Animation of balloon valvotomy
of pulmonary artery with pulmonary stenosis.
Surgery may be necessary in a variety of conditions associated
with pulmonary valve dysplasia.
Balloon valvuloplasty may not be able to open a dysplastic pulmonary
valve.
Severe right ventricular hypoplasia may be associated with critical
pulmonary stenosis requiring univentricular palliation, which
is a staged repair ultimately requiring a Fontan (right atrial
to pulmonary artery) modification.
Consultations: Pediatric cardiology consultation precedes consultation
with a cardiothoracic surgeon.
Pulmonary valve atresia or critical pulmonary stenosis with
an inadequate right ventricle requires a shunt (usually modified
Blalock-Taussig or central shunt) after the ductus arteriosus
is kept patent pharmacologically with prostaglandin E1.
Definitive repair may not be possible if the right ventricle
is hypoplastic, requiring a single ventricular palliation, such
as the Fontan procedure, or variation, such as a direct right
atrial appendage to main pulmonary artery anastomosis.
Activity: A prudent philosophy is to allow patients to limit
their own activity according to personal tolerance.
MEDICATION
No medications are useful in isolated valvar pulmonary stenosis.
Patients with CHF may benefit from anticongestive therapy. Cyanotic
patients may benefit from oxygen and prostaglandin E1. Patients
with cyanosis from a large right-to-left shunt require a definitive
surgical procedure.
Drug Category: Prostaglandins -- Alprostadil (Prostaglandin
E1, PGE1) is used for treatment of ductal-dependent cyanotic
congenital heart disease, which is caused by decreased pulmonary
blood flow. It acts as a smooth muscle relaxer and maintains
patency of the ductus arteriosus when a cyanotic lesion (ie,
critical pulmonary stenosis or atresia) or an interrupted aortic
arch presents in a newborn. It is more effective in premature
infants than in mature infants.
| Drug name |
Alprostadil (Prostin VR) -- First-line medication
used as palliative therapy to temporarily maintain patency
of the ductus arteriosus before surgery. Produces vasodilation
and increases cardiac output. Also inhibits platelet aggregation
and stimulates intestinal and uterine smooth muscle. Used
for suspected critical pulmonary stenosis when presentation
includes cyanosis, and with a ductal-dependent lesion (eg,
pulmonary atresia variants, coarctation of the aorta, interrupted
aortic arch). Each 1-mL ampule contains 500 mcg/mL. |
| Pediatric Dose |
0.01 mcg/kg/min; up to 0.4 mcg/kg/min IV |
| Contraindications |
Documented hypersensitivity; hyaline membrane
disease, respiratory distress syndrome |
| Interactions |
Limited data exist; caution with concurrent
use of antiplatelet drugs or anticoagulants |
| Pregnancy C |
Safety for use during pregnancy has not been
established. |
| Precautions |
Adverse effects and toxicity
include apnea, seizures, fever, hypotension, leukocytosis,
fever and pulmonary overcirculation; neonates usually are
intubated prophylactically because of potential risk of
apnea (10-12%); prolonged use occasionally is necessary
(in hypoplastic left heart syndrome transplant candidates)
and may be associated with third spacing of fluid; monitor
blood oxygenation and arterial pressure |
| Drug Category: |
Antibiotics, prophylactic -- Antibiotic prophylaxises
is given to patients before undergoing procedure that may
cause bacteremia . |
| Drug Name |
Amoxicillin (Amoxil, Trimox) -- Interferes
with synthesis of cell wall mucopeptides during active multiplication
resulting in bactericidal activity against susceptible bacteria.
Used as prophylaxis in minor procedures. |
| Adult Dose |
2 g PO 1 h before procedure; alternatively,
3 g PO 1 h before procedure, followed by 1.5 g 6 h after
initial dose |
| Pediatric Dose |
50 mg/kg 1 h PO before procedure; not to exceed
2 g/dose |
| Contraindications |
Documented hypersensitivity |
| Interactions |
Reduces the efficacy of oral contraceptives
|
| Pregnancy B |
Usually safe but benefits must outweigh the
risks. |
| Precautions |
Adjust dose in renal impairment |
| Drug Name |
Ampicillin (Marcillin, Omnipen) -- For prophylaxis
in patients undergoing dental, oral, or respiratory tract
procedures. Coadministered with gentamicin for prophylaxis
in gastrointestinal or genitourinary procedures. |
| Adult Dose |
2 g IV/IM 30 min before procedure |
| High-risk patients: |
2 g ampicillin IV/IM plus 1.5 mg/kg gentamicin
30 min before procedure, followed 6 h later by 1 g ampicillin
IV/IM or 1 g amoxicillin PO |
| Pediatric Dose |
50-mg/kg IV/IM 30 min before procedure; not
to exceed 2 g/dose
High-risk patients: 50 mg/kg IV/IM ampicillin plus gentamicin
1.5 mg/kg 30 min before procedure, followed 6 h later by
ampicillin 25 mg/kg IV/IM or amoxicillin 25 mg/kg PO |
| Contraindications |
Documented hypersensitivity |
| Interactions |
Probenecid and disulfiram elevate levels;
allopurinol decreases ampicillin effects and has additive
effects on ampicillin rash; may decrease effects of oral
contraceptives |
| Pregnancy B |
Usually safe but benefits must outweigh the
risks. |
| Precautions |
Adjust dose in renal failure; evaluate rash
and differentiate from hypersensitivity reaction |
| Drug Name |
Clindamycin (Cleocin) -- Used in penicillin-allergic
patients undergoing dental, oral, or respiratory tract procedures.
Useful for treatment against streptococcal and most staphylococcal
infections. |
| Adult Dose |
600 mg PO/IV 1 h before procedure and 150
mg PO/IV 6 h after first dose |
| Pediatric Dose |
20 mg/kg PO 1 h or 20 mg/kg IV 30 min before
procedure; not to exceed 600 mg/dose |
| Contraindications |
Documented hypersensitivity; regional enteritis,
ulcerative colitis, hepatic impairment, antibiotic-associated
colitis |
| Interactions |
Increases duration of neuromuscular blockade,
induced by tubocurarine and pancuronium; erythromycin may
antagonize effects of clindamycin; antidiarrheals may delay
absorption of clindamycin |
| Pregnancy B |
Usually safe but benefits must outweigh the
risks. |
| Precautions |
Adjust dose in severe hepatic dysfunction;
no adjustment necessary in renal insufficiency; associated
with severe and possibly fatal colitis |
| Drug Name |
Gentamicin (Garamycin) -- Aminoglycoside antibiotic
for gram-negative coverage. Used in combination with both
an agent against gram-positive organisms and one that covers
anaerobes. Used in conjunction with ampicillin or vancomycin
for prophylaxis in GI or genitourinary procedures. |
| Adult Dose |
1.5 mg/kg IV; not to exceed 120 mg/dose; administer
with 1-2 g ampicillin 30 min before procedure; not to exceed
80 mg |
| Pediatric Dose |
2 mg/kg IV; not to exceed 120 mg/dose, with
ampicillin (50 mg/kg IV; not to exceed 2 g/dose) 30 min
before procedure |
| Contraindications |
Documented hypersensitivity; non–dialysis-dependent
renal insufficiency |
| Interactions |
Coadministration with other aminoglycosides,
cephalosporins, penicillins, and amphotericin B may increase
nephrotoxicity; aminoglycosides enhance effects of neuromuscular
blocking agents (thus, prolonged respiratory depression
may occur); coadministration with loop diuretics may increase
auditory toxicity of aminoglycosides; possible irreversible
hearing loss of varying degrees may occur (monitor regularly)
|
| Pregnancy C |
Safety for use during pregnancy has not been
established. |
| Precautions |
Narrow therapeutic index (not intended for
long-term therapy); caution in renal failure (not on dialysis),
myasthenia gravis, hypocalcemia, and conditions that depress
neuromuscular transmission; adjust dose in renal impairment
|
| Drug Name |
Vancomycin (Vancocin) -- Potent antibiotic
directed against gram-positive organisms and active against
Enterococcus species. Useful in the treatment of septicemia
and skin structure infections. Indicated for patients who
cannot receive, or have failed to respond to, penicillins
and cephalosporins or have infections with resistant staphylococci.
Use CrCl to adjust dose in patients with renal impairment.
Used in conjunction with gentamicin for prophylaxis in penicillin-allergic
patients undergoing gastrointestinal or genitourinary procedures.
|
| Adult Dose |
Dental, oral or upper respiratory tract surgery:
1 g IV, infused over 1 h, 1 h before procedure |
| GI/GU procedures: |
1 g IV plus gentamicin 1.5 mg/kg IV infused
over 1 h, 1 h before surgery |
| Pediatric Dose |
Dental, oral, or upper respiratory tract surgery:
20 mg/kg IV, infused over 1 h, 1 h before procedure |
| Contraindications |
Documented hypersensitivity |
| Interactions |
Erythema, histaminelike flushing and anaphylactic
reactions may occur when administered with anesthetic agents;
taken concurrently with aminoglycosides, risk of nephrotoxicity
may increase more than that in aminoglycoside monotherapy;
effects in neuromuscular blockade may be enhanced when coadministered
with nondepolarizing muscle relaxants |
| Pregnancy C |
Safety for use during pregnancy has not been
established. |
| Precautions |
Caution in renal failure, neutropenia; red
man syndrome caused by too rapid IV infusion (dose given
over a few min) but rarely occurs when dose given as 2-h
administration or as PO or IP administration; red man syndrome
is not an allergic reaction |
| Drug Name |
Cefazolin (Ancef) -- First-generation semisynthetic
cephalosporin that arrests bacterial cell wall synthesis,
inhibiting bacterial growth. Primarily active against skin
flora, including Staphylococcus aureus. |
| Adult Dose |
1 g IV/IM within 30 min before procedure |
| Pediatric Dose |
25 mg/kg IV/IM within 30 min before procedure;
not to exceed 1 g/dose |
| Contraindications |
Documented hypersensitivity |
| Interactions |
Probenecid prolongs effect of cefazolin; coadministration
with aminoglycosides, may increase renal toxicity; may yield
false-positive urine dipstick test result for glucose |
| Pregnancy B |
Usually safe but benefits must outweigh the
risks. |
| Precautions |
Adjust dose in renal impairment; superinfections
and promotion of nonsusceptible organisms may occur with
prolonged use or repeated therapy |
| Drug Name |
Cephalexin (Keflex)First-generation cephalosporin arrests
bacterial growth by inhibiting bacterial cell wall synthesis.
Bactericidal activity against rapidly growing organisms.
Primary activity against skin flora and used for skin infections
or prophylaxis in minor procedures |
| Adult Dose |
2 g PO 1 h before procedure |
| Pediatric Dose |
50 mg/kg PO 1 h before procedure; not to exceed
2 g/dose |
| Contraindications |
Documented hypersensitivity |
| Interactions |
Coadministration with aminoglycosides increase
nephrotoxic potential |
| Pregnancy B |
Usually safe but benefits must outweigh the
risks. |
| Precautions |
Adjust dose in renal impairment |
| Drug Name |
Cefadroxil (Duricef) |
| |
First-generation cephalosporin arrests bacterial
growth by inhibiting bacterial cell wall synthesis. Bactericidal
activity against rapidly growing organisms. Primary activity
against skin flora and used for skin infections or prophylaxis
in minor procedures. |
| Adult Dose |
2 g PO 1 h before procedure |
| Pediatric Dose |
50 mg/kg PO 1 h before procedure; not to exceed
2 g/dose |
| Contraindications |
Documented hypersensitivity |
| Interactions |
Coadministration with furosemide or aminoglycosides
may increase nephrotoxicity; probenecid prolongs effects
|
| Pregnancy B |
Usually safe but benefits must outweigh the
risks. |
| Precautions |
Adjust dose in renal impairment; superinfections,
and promotion of nonsusceptible organisms may occur with
prolonged use or repeated therapy |
| Drug Name |
Azithromycin (Zithromax) Inhibits bacterial
growth, possibly by blocking dissociation of peptidyl tRNA
from ribosomes causing RNA-dependent protein synthesis to
arrest. |
| Adult Dose |
500 mg PO 1 h before procedure |
| Pediatric Dose |
15 mg/kg PO 1 h before procedure; not to exceed
500 mg/dose |
| Contraindications |
Documented hypersensitivity; hepatic impairment;
not to administer with pimozide |
| Interactions |
May increase toxicity of theophylline, warfarin,
and digoxin; effects are reduced with coadministration of
aluminum and/or magnesium antacids; nephrotoxicity and neurotoxicity
may occur when coadministered with cyclosporine |
| Pregnancy B |
B - Usually safe but benefits must outweigh
the risks |
| Precautions |
Bacterial or fungal overgrowth may result
with prolonged antibiotic use; may increase hepatic enzymes
and cholestatic jaundice; caution in patients with impaired
hepatic function, prolonged QT intervals, or pneumonia;
caution in elderly patients and in patients who are hospitalized
or debilitated |
| Drug Name |
Clarithromycin (Biaxin) -- Inhibits bacterial
growth, possibly by blocking dissociation of peptidyl tRNA
from ribosomes causing RNA-dependent protein synthesis to
arrest. |
| Adult Dose |
500 mg PO 1 h before procedure |
| Pediatric Dose |
15 mg/kg PO 1 h before procedure; not to exceed
500 mg/dose |
| Contraindications |
Documented hypersensitivity; coadministration
of pimozide |
| Interactions |
Toxicity increases with coadministration of
fluconazole, astemizole, and pimozide; clarithromycin effects
decrease and GI adverse effects may increase with coadministration
of rifabutin or rifampin; may increase toxicity of anticoagulants,
cyclosporine, tacrolimus, digoxin, omeprazole, carbamazepine,
ergot alkaloids, triazolam, and HMG CoA-reductase inhibitors;
cardiac arrhythmias may occur with coadministration of cisapride;
plasma levels of certain benzodiazepines may increase, prolonging
CNS depression; arrhythmias and increase in QTc intervals
occur with disopyramide; coadministration with omeprazole
may increase plasma levels of both agents |
| Pregnancy C |
Safety for use during pregnancy has not been
established. |
| Precautions |
Coadministration with ranitidine or bismuth
citrate is not recommended with CrCl <25 mL/min; give
half dose or increase dosing interval if CrCl <30 mL/min;
diarrhea may be sign of pseudomembranous colitis; superinfections
may occur with prolonged or repeated antibiotic therapies
|
| |
|
FOLLOW-UP
Further Inpatient Care:
Patients with mild valvar pulmonary stenosis (<25 mmHg)
do not experience an increase in gradient, nor do they require
any treatment.
Choice of management of patients with gradients of 40-49 mmHg
remains a matter of debate.
Patients with a gradient greater than or equal to 50 mm Hg should
have valvotomy or valvuloplasty. If valvotomy or valvuloplasty
is required in a child, reoperation rarely is necessary.
The neonate with critical pulmonary stenosis requires special
consideration. Critical pulmonary stenosis may present with
near pulmonary atresia (cyanotic lesion) with a small and often
inadequate right ventricle. These patients survive because of
a patent ductus arteriosus.
Patients with severe or symptomatic infundibular or supravalvar
pulmonary stenosis require surgical intervention.
Pulmonary valve atresia or critical pulmonary stenosis with
an inadequate right ventricle may require a shunt (usually a
modified Blalock-Taussig or central shunt) if it is deemed impossible
to "puncture" the pulmonary valve and balloon dilate.
Throughout the procedure, the ductus arteriosus is kept patent
pharmacologically with prostaglandin E1.
Definitive repair may not be possible if the right ventricle
is hypoplastic, requiring a single ventricular palliation, such
as the Fontan procedure, or a variation of this. The Fontan
procedure is a direct right atrial appendage to main pulmonary
artery anastomosis.
Frequently, the main and branch pulmonary arteries require augmentation
prior to a Fontan, especially if a prior systemic to pulmonary
artery shunt was performed (modified Blalock-Taussig shunt).
This is not performed for pure valvar pulmonary stenosis.
Balloon valvuloplasty has become an accepted alternative to
surgery for valvar stenosis.
Balloon dilation avoids a potentially painful operation and
a long postoperative recovery, and at the same time offers substantial
cost savings. However, such advantages are meaningless if the
safety of the interventional procedure does not match or surpass
the results of conventional surgery.
Further Outpatient Care:
Physical activity should be normal.
Most patients with PS are given subacute bacterial endocarditis
(SBE) prophylaxis.
Opinions differ as to the need for SBE prophylaxis recommendations
for valvar pulmonary stenosis because of the extremely low incidence
of pulmonary valve endocarditis in this relatively large subpopulation.
Transfer:
Transfer patients with symptomatic pulmonary stenosis to a
tertiary care center offering pediatric cardiology and pediatric
cardiothoracic surgery.
Complications:
One complication with the acute palliation for severe pulmonary
stenosis involves the hypercontractile residual obstructing
muscular hypertrophy in the infundibulum.
This phenomenon of infundibular obstruction after valvar stenosis
repair by surgery or valvuloplasty has led to the designation
of a "suicide right ventricle."
Beta-blockers and volume replacement are used to treat this
condition, which occurs more frequently in older patients with
long-standing pulmonary stenosis.
Late atrial arrhythmias
Persistent repolarization abnormalities
Prognosis:
Mild valvar pulmonary stenosis usually does not progress, but
the moderate-to-severe disease does tend to progress.
After relief of the stenosis, the condition does not recur,
and right ventricular hypertrophy will regress.
Following balloon or surgical valvulotomy, the outcome generally
is excellent. Probability of survival is similar to that of
the general population, and the vast majority of patients are
asymptomatic.
Patient Education:
Reassure patients and parents of those with mild valvar pulmonary
stenosis that this condition is not related to, or associated
with, coronary artery disease, dysrhythmia, or sudden death.
Insurability may become a factor in obtaining further care.
Patients are no more at risk for disastrous health consequences
than the usual population.
Provided the patient is asymptomatic, acyanotic, and has mild
valvar pulmonary stenosis by initial Doppler echocardiography,
a yearly screening examination and electrocardiogram would be
prudent follow-up care.
If no significant change in the evaluation is present a few
years after the initial evaluation, the patient can be reasonably
discharged for follow-up care over extended periods of 3-5 years.
MISCELLANEOUS
Medical/Legal Pitfalls:
Failure to exclude associated congenital anomalies and detect
the presence of cyanosis or a ductal-dependent lesion is a major
error.
Failure to diagnose a more serious congenital heart defect,
such as tetralogy of Fallot, could yield disastrous consequences.
Acyanotic patients with tetralogy of Fallot and mild right ventricular
outflow tract obstruction may have a similar presentation and
physical examination.
Tetralogy of Fallot is a lesion that is surgically correctable
and can be corrected safely, even in the neonatal period.
A "tet spell," or hypercyanotic spell, is potentially
lethal, frequently aborted with simple skills, and can occur
in previously pink tets.
Echocardiography can reliably confirm the precise diagnosis
and differentiate between valvar pulmonary stenosis and tetralogy
of Fallot.
Echocardiography should not be withheld if any suspicion of
a more complex anatomy exists.